| World Journal of Nephrology and Urology, ISSN 1927-1239 print, 1927-1247 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Nephrol Urol and Elmer Press Inc |
| Journal website https://wjnu.elmerpub.com |
Review
Volume 15, Number 2, April 2026, pages 26-41
Endocrine Disorders in Adults With End-Stage Renal Disease on Dialysis: A Review
Vincenzo Barberaa, f, g , Lorenzo D’Eliaa, Luca Di Lullob, Paola Peverinib, Aldo Francullib, f, Pasquale Saporitob, Andrea Dello Strologob, c, Deborah Di Vicoa, Giovanni Otrantoa, Annalisa Villania, Antonio Bellasid, e, f, g
aDepartment of Nephrology and Dialysis, Ospedale L. Parodi Delfino, Azienda USL Roma 5, Colleferro, Italy
bDepartment of Nephrology and Dialysis, Ospedale dei Castelli, Azienda USL Roma 6, Ariccia, Italy
cDepartment of Translational Medicine and Surgery, Universita Cattolica del Sacro Cuore, Rome, Italy
dService of Nephrology, Ospedale Regionale di Lugano, Ospedale Civico, Ente Ospedaliero Cantonale Via Tessarete 46, CH-6903 Lugano, Switzerland
eFaculty of Biomedical Sciences, Universita della Svizzera italiana (USI), 6900 Lugano, Switzerland
fThese authors contributed equally to this work.
gCorresponding Authors: Vincenzo Barbera, Department of Nephrology and Dialysis, Ospedale L. Parodi Delfino, Azienda USL Roma 5, Colleferro, Italy; Antonio Bellasi, Service of Nephrology, Ospedale Regionale di Lugano, Ospedale Civico, Ente Ospedaliero Cantonale, Via Tessarete 46, CH-6903 Lugano, Switzerland
Manuscript submitted January 16, 2026, accepted February 12, 2026, published online March 23, 2026
Short title: Endocrine Disorders in Dialyzed ESRD Adults
doi: https://doi.org/10.14740/wjnu1036
| Abstract | ▴Top |
Background: Endocrine dysfunctions are frequent yet underrecognized complications of end-stage renal disease (ESRD), with major implications for prognosis, quality of life, and treatment. This review provides an updated synthesis of endocrine alterations in ESRD, emphasizing clinical impact, dialysis modality differences, and research needs.
Methods: A systematic search of PubMed and EMBASE identified studies published between January 1975 and January 2026, including observational studies, randomized controlled trials (RCTs), systematic reviews, and international guidelines. Evidence was synthesized across major pathways in ESRD: hypothalamic-pituitary-gonadal axis, prolactin, thyroid function, vitamin D-parathyroid hormone metabolism, and adrenal function. Particular emphasis was placed on pivotal RCTs (EVOLVE, PRIMO, OPERA, phosphodiesterase type 5 inhibitor studies) and key guidelines statements (Endocrine Society, American Thyroid Association (ATA), European Menopause and Andropause Society (EMAS), Kidney Disease: Improving Global Outcomes (KDIGO), European Renal Association–European Dialysis and Transplantation Association (ERA-EDTA), American Urological Association (AUA)).
Results: Hypogonadism affects up to two-thirds of male dialysis patients, contributing to cardiovascular risk, anemia, and mortality; in women on dialysis, ovarian failure drives infertility and sexual dysfunction. Hyperprolactinemia is highly prevalent and exacerbates gonadal dysfunction, with potential cardiovascular relevance. Thyroid alterations include “low-T3 syndrome”, an independent predictor of mortality, and increased risk of subclinical and overt hypothyroidism. Chronic kidney disease-mineral and bone disorders contributes to bone disease, vascular calcification, and adverse outcomes. Dialysis modality (hemodialysis vs. peritoneal dialysis) influences endocrine stability and quality of life.
Conclusions: Endocrine disorders in ESRD are prognostically relevant and require systematic screening, tailored management, and multidisciplinary care. Current evidence is predominantly observational, underscoring the urgent need for randomized trials to establish optimal therapeutic strategies and clarify their impact on survival and cardiovascular outcomes.
Keywords: Endocrine alterations; End-stage renal disease; Hyperprolactinemia; Hypogonadism
| Introduction | ▴Top |
Endocrine alterations in patients with chronic kidney disease (CKD) represent an area of growing clinical and scientific interest, as highlighted by seminal reviews that have outlined the main pathophysiological mechanisms and the clinical implications of these dysfunctions [1, 2]. In this context, diabetes mellitus, a primary endocrine disorder, represents the leading cause of CKD and end-stage renal disease (ESRD) worldwide, accounting for a substantial proportion of patients requiring renal replacement therapy (RRT) [3].
ESRD is a complex and multifactorial clinical condition characterized by a progressive decline in kidney function, ultimately requiring renal replacement therapies such as hemodialysis (HD) or peritoneal dialysis (PD) [4]. Despite the technological and organizational advances of recent decades, which have significantly improved survival and management of well-recognized complications such as anemia and chronic kidney disease-mineral and bone disorders (CKD-MBD), patient’s quality of life remains severely compromised [5–8]. A substantial proportion of this clinical burden is attributable to endocrine dysfunction (Fig. 1) which, although highly prevalent, is often understudied in routine clinical practice.
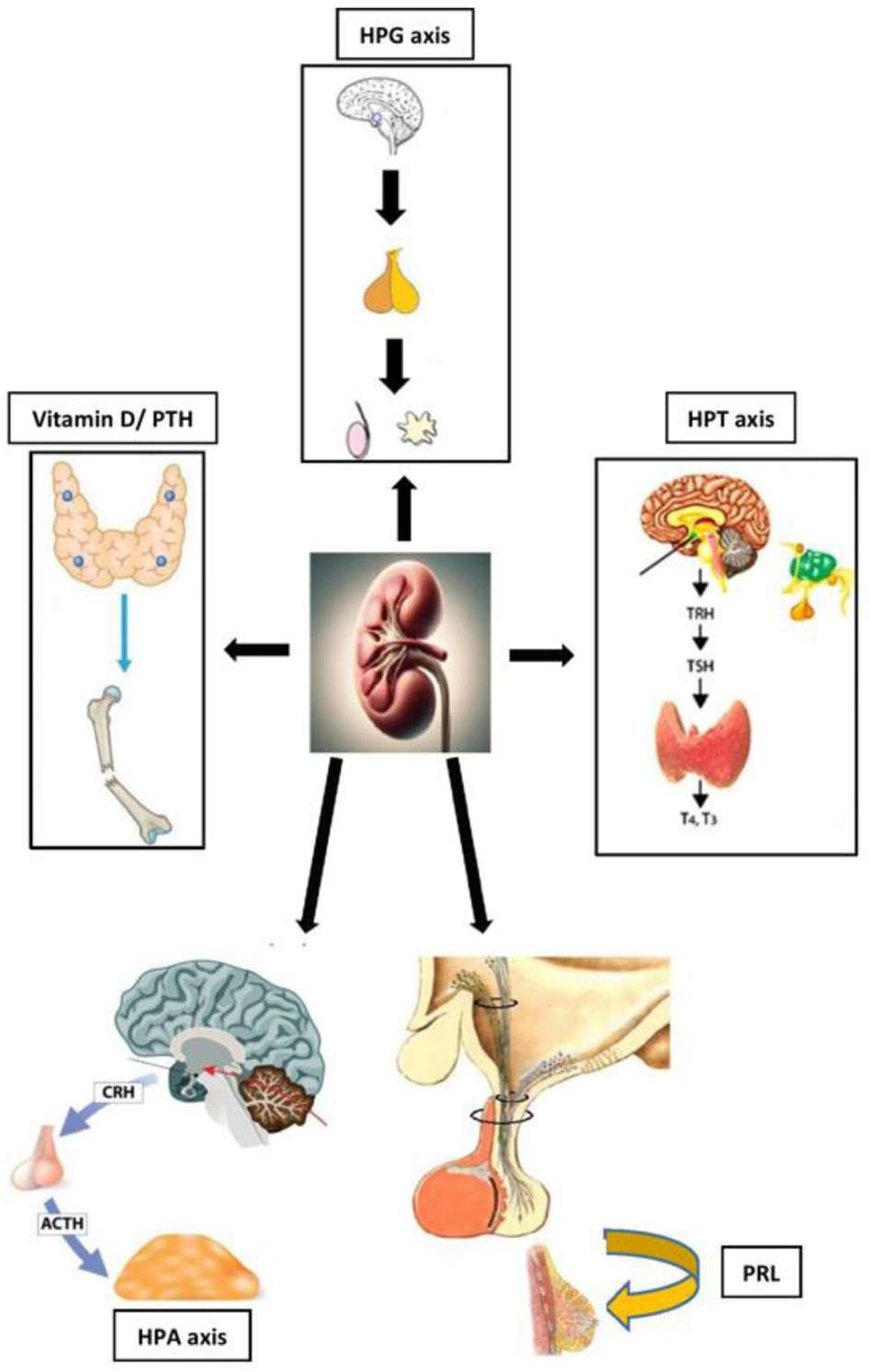 Click for large image | Figure 1. Schematic representation of the interplay between kidney dysfunction in end-stage renal disease (ESRD) and major endocrine axes: hypothalamic–pituitary–gonadal (HPG), hypothalamic–pituitary–thyroid (HPT), hypothalamic–pituitary–adrenal (HPA), prolactin (PRL), and vitamin D/parathyroid hormone (PTH). |
Among the most frequent alterations are the dysfunction of hypothalamic–pituitary–gonadal (HPG) axis, manifesting as hypogonadism in both men and women; abnormalities of hypothalamic–pituitary–thyroid (HPT) axis, including the “low-T3” syndrome, hypothyroidism and, less commonly, hyperthyroidism; and hyperprolactinemia due to reduced renal clearance [9, 10]. These disturbances are not merely “biochemical epiphenomena” of uremia, but clinically relevant conditions that directly affect patient’s daily well-being, sexual and reproductive function, nutritional status, and overall survival [11, 12].
In male dialysis patients, low testosterone levels are associated with erectile dysfunction, sarcopenia, erythropoiesis-stimulating agents (ESAs)-resistant anemia, and increased cardiovascular mortality [13–15]. In women, hypogonadism manifests as amenorrhea, infertility, reduced libido, and impaired quality of life [16, 17]. Similarly, reduced free triiodothyronine (FT3) has been recognized as an independent predictor of mortality in dialysis patients [18], while hyperprolactinemia contributes to worsening gonadal dysfunction, impaired sexual function, and metabolic abnormalities [9].
These data underscore that the endocrine profile is not only a marker of CKD severity but also a prognostic determinant and a potential therapeutic target. An additional point of interest concerns the differences between dialysis modalities (HD and PD). Indeed, it has been observed that the choice of dialysis modality may differentially affect the hormonal profile and patients’ quality of life, with potential implications for selecting the most appropriate RRT [19, 20]. Moreover, kidney transplantation, whereas available, remains the only truly curative option capable of normalizing most endocrine abnormalities [21].
In light of these considerations, this review aims to provide an updated and critical overview of the major endocrine alterations in patients affected by ESRD undergoing RRT. We will discuss the underlying pathophysiology, clinical manifestations, differences between HD and PD, and currently available therapeutic options, with particular reference to international guidelines (Endocrine Society, ATA, European Menopause and Andropause Society (EMAS), Kidney Disease: Improving Global Outcomes (KDIGO), ERA-EDTA, American Urological Association (AUA)). The ultimate goal is to promote a multidisciplinary approach integrating endocrinological and nephrological expertise, acknowledging the central role of hormonal dysfunction in determining prognosis and quality of life in this complex patient population.
| Methods | ▴Top |
Following PRISMA guidelines (Fig. 2), we conducted a comprehensive literature search across PubMed and EMBASE between January 1975 and January 2026 using the following keywords, either individually or in combination: end-stage renal disease, chronic kidney disease, hemodialysis, peritoneal dialysis, endocrine dysfunction, hypogonadism, prolactin (PRL), hyperprolactinemia, thyroid disorders, hypothyroidism, hyperthyroidism, parathyroid hormone, vitamin D, adrenal function, fertility, sexual dysfunction. Only studies conducted in adult populations and published in English/Italian language were considered. We included original studies (randomized controlled trials (RCTs), prospective and observational studies), meta-analyses, systematic reviews, international guidelines, and consensus statements from major scientific societies (KDIGO, EMAS, ERA-EDTA). Studies limited to single case reports, abstracts lacking complete data, and articles not available in English or Italian were excluded. Article selection was conducted in two phases: an initial screening of titles and abstracts, followed by full-text analysis. Particular attention was given to studies comparing HD and PD, as well as those evaluating kidney transplantation as a comparator variable. For clinical practice, we also considered the recommendations of the Endocrine Society and the AUA for the diagnosis and management of androgen deficiency [22], as well as recent literature highlighting the impact of female sexual dysfunction in patients with ESRD [23]. The aim was not to perform a qualitative meta-analysis, but rather to provide an integrated review of the major endocrine alterations in ESRD (Table 1), with emphasis on their clinical implications and on multidisciplinary therapeutic perspectives.
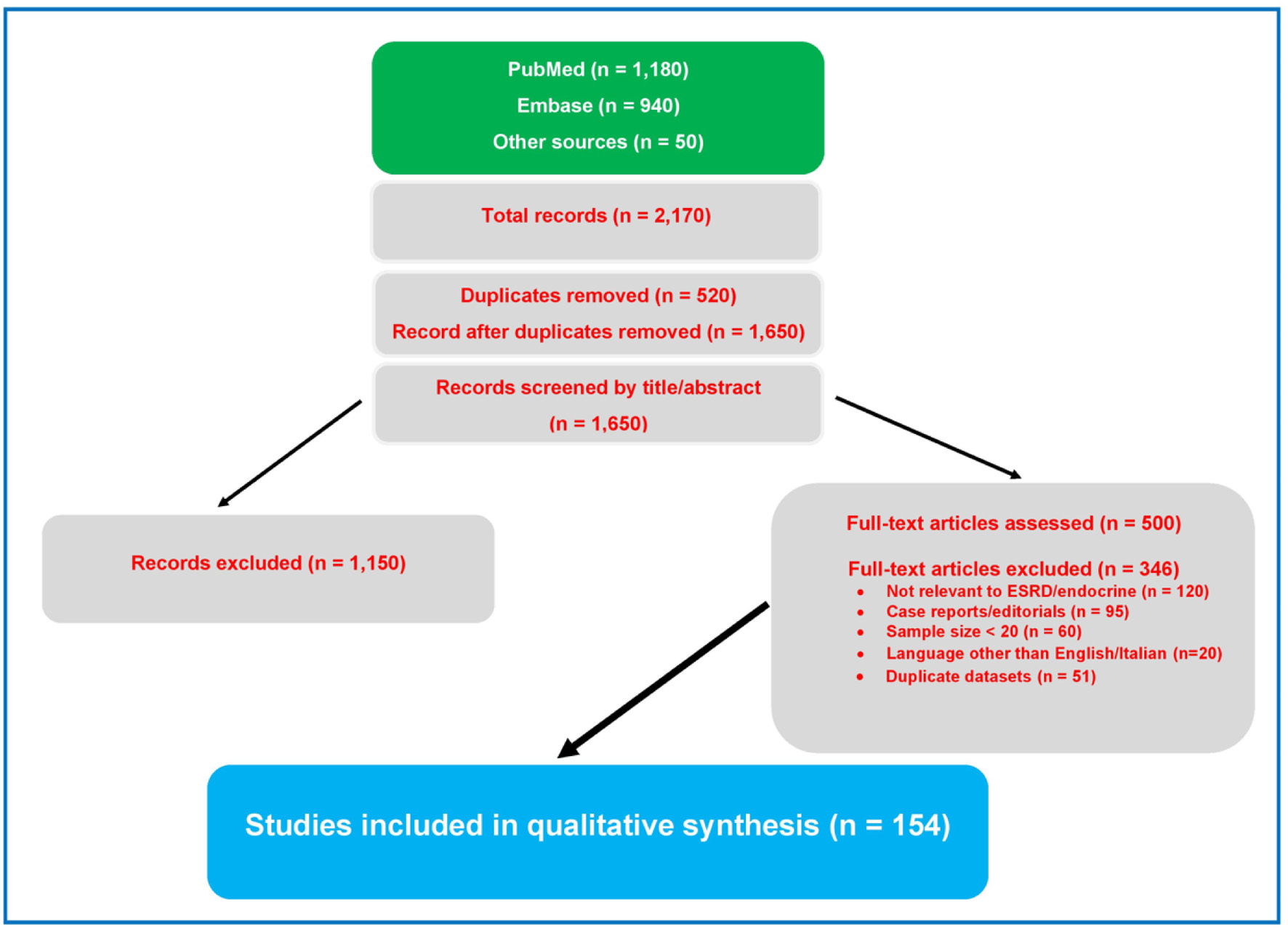 Click for large image | Figure 2. PRISMA flow diagram of the study selection process. From 2,170 records initially identified, 1,650 remained after duplicate removal. Following title/abstract screening and full text assessment, 154 studies were included in the qualitative synthesis. |
 Click to view | Table 1. Main Endocrine Alterations in Patients With ESRD |
| Results and Discussion | ▴Top |
HPG axis
Male sex
Hypogonadism represents one of the most clinically relevant endocrine complications in patients with ESRD, with reported prevalence ranging from 27% to 66%, depending on diagnostic criteria and study population considered [24, 25]. This wide variability reflects not only methodological differences in clinical cohorts but also patient heterogeneity in terms of age, metabolic comorbidities, and duration of dialysis therapy. The pathophysiology of male hypogonadism in ESRD is complex and multifactorial. A key mechanism is the uremic milieu—including retained solutes (“uremic toxins”) and inflammatory mediators—which has been associated with impaired hypothalamic gonadotropin-releasing hormone (GnRH) pulsatility and reduced pituitary gonadotropin release (luteinizing hormone (LH)/follicle-stimulating hormone (FSH)), thereby contributing to functional hypogonadotropic hypogonadism. This hypogonadotropic pattern in men with ESRD contrasts with the predominantly hypergonadotropic hypogonadism observed in women of reproductive age on dialysis, in whom elevated LH and FSH levels mainly reflect impaired ovarian steroidogenesis and disturbed feedback rather than preserved GnRH–LH pulsatility. In other words, uremia exerts a central inhibitory effect on GnRH secretion in both sexes, but in women the coexistence of reduced negative and positive estradiol feedback and possible reduced gonadotropin clearance leads to chronically elevated gonadotropin levels despite anovulation and luteal insufficiency. Importantly, uremia does not selectively affect GnRH alone; rather, it induces a broader hypothalamic–pituitary dysregulation (e.g., dopaminergic control of PRL and altered thyrotropic hormone releasing hormone (TRH)/thyroid-stimulating hormone (TSH) dynamics), as discussed in the respective sections [26–28]. This is compounded by hyperprolactinemia, which exerts an additional suppressive effect on the hypothalamic-pituitary axis [29, 30]. Furthermore, the chronic inflammatory state typical of uremia, with increased levels of pro-inflammatory cytokines (interleukin (IL)-6, tumor necrosis factor (TNF)-α) and oxidative stress, impairs Leydig cell function, thereby reducing testosterone synthesis [31, 32]. Concomitant metabolic factors such as diabetes mellitus, obesity, and metabolic syndrome further aggravate gonadal dysfunction through mechanisms of insulin resistance and peripheral hyperestrogenism [33, 34].
The clinical consequences of hypogonadism in dialysis patients extend well beyond reduced libido and erectile dysfunction. Androgen deficiency is associated with sarcopenia, decreased muscle strength, increased visceral fat mass, reduced responsiveness to ESAs, and worsening of anemia [35]. From a prognostic perspective, several studies have demonstrated that low total testosterone levels (< 300 ng/dL) are an independent predictor of both cardiovascular and all-cause mortality (Fig. 3), with an increased risk of up to 30% in dialysis patients [36, 37].
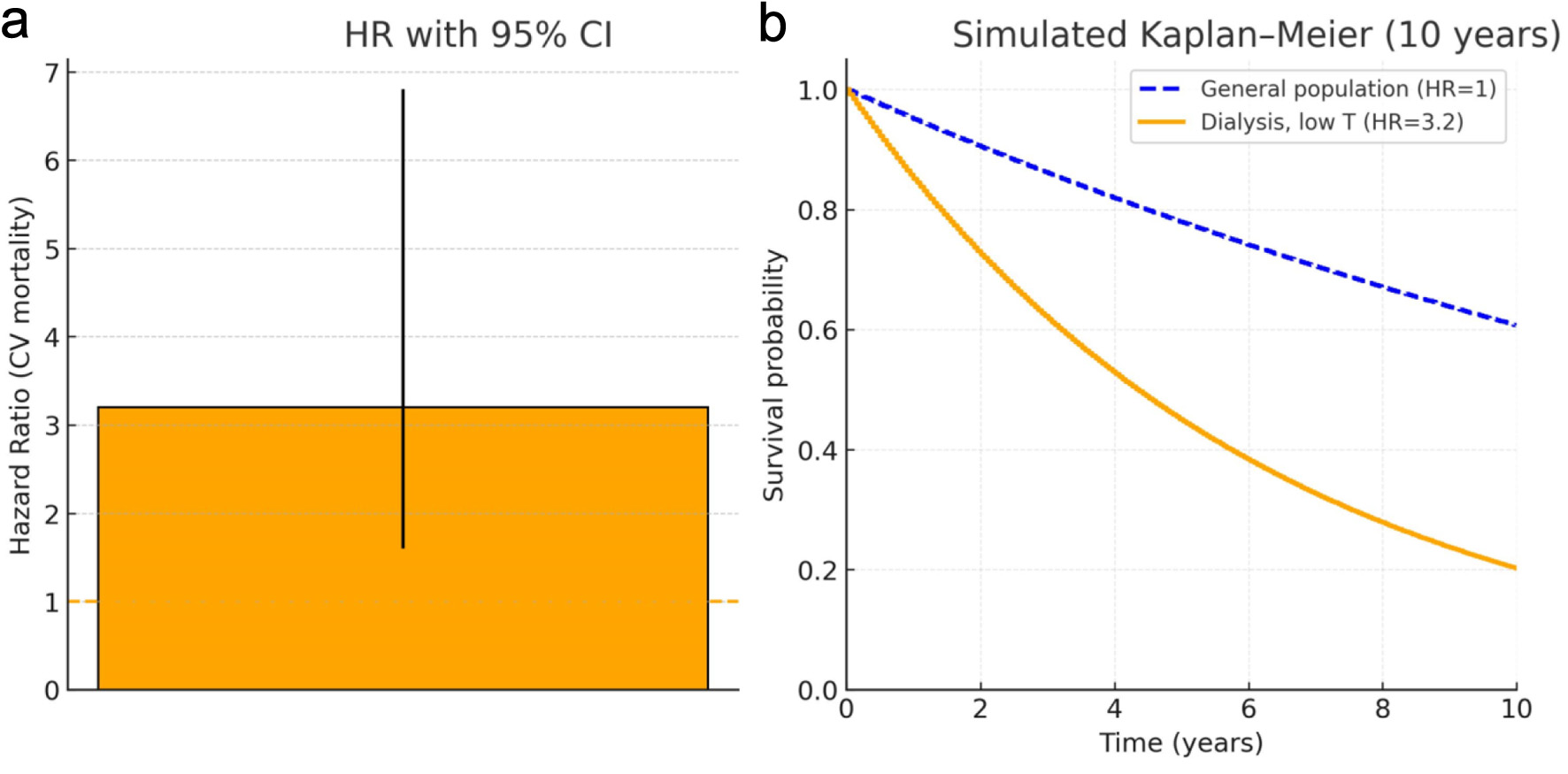 Click for large image | Figure 3. Survival impact of low testosterone in dialysis patients. Low testosterone was defined as total testosterone < 10 nmol/L (< 288 ng/dL), according to the original criteria used by Carrero et al [37]. (a) Hazard ratio (HR) for cardiovascular mortality associated with low testosterone levels in dialysis patients, according to Carrero et al [37]. Reported HR = 3.2; 95% CI: 1.6–6.8 (Adapted/reproduced from Carrero et al [37] with permission where required). (b) Simulated Kaplan–Meier survival curves over 10 years comparing the general population (hazard ratio (HR) = 1, dashed blue line) and dialysis patients with low testosterone (HR = 3.2, solid orange line; derived from Carrero et al [37]). Curves are illustrative and generated from an exponential survival model using the reported HR value. |
Diagnosis requires an integrated approach. Clinical evaluation should be supported by validated questionnaires such as the International Index of Erectile Function (IIEF-5), combined with measurement of total testosterone in at least two morning samples [38]. In borderline cases, assessment of free testosterone, either calculated on the basis of sex hormone-binding globulin (SHBG) or measured directly, may be informative. Gonadotropins (LH, FSH) measurement helps differentiate hypogonadotropic from hypergonadotropic forms, with important clinical implications [39].
The treatment of male hypogonadism in ESRD is complex and requires a careful balance of benefits and risks. Phosphodiesterase type 5 inhibitors (PDE-5i), such as sildenafil and tadalafil, represent the first-line therapy for erectile dysfunction and have shown efficacy even in dialysis patients. Key evidence for endocrine practice includes clinical trials on the use of PDE-5i in dialysis patients with erectile dysfunction: RCTs have demonstrated significant improvements in IIEF scores and quality of life with sildenafil and vardenafil, with a favorable safety profile. These findings support the use of PDE-5i as first-line therapy in ESRD patients, in alignment with current recommendations for erectile dysfunction management in the presence of hypogonadism [41].
Regarding testosterone replacement therapy (TRT), the evidence base in dialysis patients remains limited but clinically important. TRT may be considered in patients with symptomatic, biochemically confirmed hypogonadism, but requires exceptionally close monitoring due to the risks of polycythemia, fluid retention, and potential worsening of heart failure. The Endocrine Society guidelines recommend restrictive indications, requiring both clinical symptoms and consistently low testosterone levels (typically < 300 ng/dL), combined with structured monitoring protocols including hematocrit, prostate-specific antigen (PSA), and cardiovascular event surveillance—aspects of particular relevance in the dialysis population [42, 43]. A recent translational review summarized the biological rationale and current gaps in hard clinical outcome data regarding TRT in CKD, emphasizing the urgent need for adequately powered prospective studies to establish optimal therapeutic strategies and clarify their impact on survival and cardiovascular outcomes. The limited RCT data specifically addressing TRT outcomes in dialysis patients underscore the significant knowledge gap in this area and support the current cautious approach to testosterone replacement in this vulnerable population.
For patients wishing to preserve fertility, alternative therapies such as clomiphene citrate, human chorionic gonadotropins (hCG), and selective estrogen modulators may represent valid options [42, 43]. These approaches warrant consideration as potential alternatives to long-term TRT given their potentially more favorable safety profiles in terms of cardiovascular and hematologic complications.
In summary, male hypogonadism in dialysis patients is not merely an endocrine disorder but also an important prognostic factor directly influencing survival and quality of life. Its management requires a multidisciplinary approach involving nephrologists, endocrinologists, and andrologists, and should be personalized according to each patients’ clinical profile. Key evidence for endocrine practice includes clinical trials on the use of PDE-5i in dialysis patients with erectile dysfunction: RCTs have demonstrated significant improvements in IIEF scores and quality of life with sildenafil and vardenafil, with a favorable safety profile [44, 45]. These findings support the use of PDE-5i as first-line therapy in ESRD patients, in alignment with current recommendations for erectile dysfunction management in the presence of hypogonadism. For TRT, the Endocrine Society guidelines recommend restrictive indications (symptoms plus consistently low testosterone) and structured monitoring (hematocrit, prostate-specific antigen (PSA), cardiovascular events), aspects of particular relevance in the dialysis population [46]. A recent translational review summarized the biological rationale and current gaps in hard clinical outcome data regarding TRT in CKD, emphasizing the urgent need for adequately powered prospective studies [47].
Female sex
Gonadal alterations in women with ESRD, although less extensively studied than in men (Table 2), represent a condition of equal relevance. Women of reproductive age undergoing dialysis treatment frequently experience menstrual disturbances, secondary amenorrhea, and infertility, reflecting significant dysfunction of the hypothalamic-pituitary-ovarian axis [16, 17]. Hormonal profiles are characterized by elevated LH and FSH levels, with reduced concentrations of estradiol and progesterone, often accompanied by hyperprolactinemia [48]. This constellation is consistent with a hypergonadotropic hypogonadism pattern, in which ovarian failure and impaired estradiol feedback (both negative and positive) lead to chronic elevation of LH/FSH despite anovulation and luteal phase defects. Classic physiological and clinical studies suggest that uremia in women blunts the normal cyclic GnRH–LH surge rather than increasing GnRH drive, and that the lack of an LH surge persists even after exogenous estradiol administration, indicating a central hypothalamic–pituitary defect. Data on GnRH levels per se in dialysis women are limited, but available evidence supports an altered pulsatile GnRH secretion and defective feedback circuitry rather than a simple up- or down-regulation of GnRH release. Clinically, female sexual dysfunction is highly prevalent and translates into impairment of overall sexual function, with a substantial impact on quality of life and reproductive health. Studies using the Female Sexual Function Index (FSFI) have demonstrated significantly lower scores in women on HD and PD compared with healthy controls, with the most pronounced effects on sexual desire, arousal, and overall satisfaction [49–51]. Interestingly, some investigations have reported greater sexual satisfaction in women on continuous ambulatory peritoneal dialysis (CAPD) compared with those on HD, likely due to greater autonomy, hemodynamic stability, and reduced variability of metabolic parameters, especially associated with PD relative to HD [52]. The consequences extend beyond sexual health: female hypogonadism contributes to increased risk of osteoporosis, sarcopenia, and cardiovascular dysfunction, thereby exacerbating the frailty of the dialysis population [53, 54].
 Click to view | Table 2. Gender Differences in Endocrine Alterations in ESRD |
As in men, the management of these disorders in women requires a personalized and multidisciplinary approach. Counseling is essential and should include clear explanations of pathophysiology, therapeutic information, and psychological support. From a therapeutic perspective, in postmenopausal women, local estrogen therapy can improve vaginal symptoms and sexual quality of life, while systemic hormone replacement therapy, preferably via the transdermal route, may be considered in selected cases, taking into account cardiovascular and thromboembolic risk [55]. The recommendations of the EMAS 2025 emphasize the importance of an individualized approach that considers age, menopausal duration, and associated comorbidities [56]. On the female side, a recent meta-analysis quantified the high prevalence of sexual dysfunction in women with ESRD and showed improvement after kidney transplantation across several FSFI domains [57]. These findings support the integration of standardized sexual function assessment into routine clinical practice and, when appropriate, multimodal counseling. In postmenopausal women with genitourinary symptoms, local estrogen therapy remains an effective option; for systemic therapy, consensus documents and international position statement (EMAS, Endocrine Society) highlight the need for individualized treatment that carefully weighs benefits and risks in the context of renal comorbidities [58, 59].
In conclusion, female hypogonadism in dialysis patients is common, underrecognized condition with significant repercussions on quality of life and prognosis. Its management requires collaboration among nephrologists, endocrinologists, and gynecologists to ensure safe and effective therapeutic strategies.
Hyperprolactinemia
Hyperprolactinemia is one of the most frequent and distinctive endocrine alterations in patients with ESRD on dialysis, with an estimated prevalence of 60–70% depending on the cohort studied [60]. Unlike the general population, in which the main causes are prolactinomas and dopamine antagonist drugs, in uremic patients this condition is essentially secondary to reduced renal clearance and central dysregulation of pituitary secretion [61].
From a pathophysiological standpoint, the kidney plays a fundamental role in PRL metabolism and degradation. In ESRD patients, the loss of this function leads to progressive accumulation of the hormone in circulation [62]. In parallel, uremia alters hypothalamic dopaminergic regulatory mechanisms, resulting in increased PRL secretion [63]. Chronic inflammation and disturbances in estrogen and androgen metabolism further contribute to sustaining hyperprolactinemia, creating a vicious cycle of endocrine dysregulation [64].
Clinical manifestations vary according to sex and age. In women of reproductive age, hyperprolactinemia is associated with oligomenorrhea and galactorrhea, compromising fertility and contributing to hypogonadotropic hypogonadism [65]. In men, it manifests as decreased libido, erectile dysfunction, and worsening testosterone deficiency [66]. In both sexes, the condition exacerbates sexual and relational quality of life, amplifying a problem already prevalent in dialysis patients [67]. Beyond the reproductive domain, several recent studies have suggested a possible association between hyperprolactinemia and cardiovascular risk in ESRD. PRL appears to exert pro-inflammatory and pro-atherogenic effects, potentially involved in the progression of endothelial dysfunction and increased arterial stiffness [68]. Although these associations are not yet fully understood, they highlight the need for greater clinical attention to this condition.
An important aspect is the response to dialysis therapy. Conventional HD is unable to significantly reduce PRL levels, which remain persistently elevated over time [69]. More intensive HD regimens, such as daily or nocturnal dialysis, may lead to partial reductions in serum levels but rarely normalize PRL concentrations. Similarly, CAPD does not appear to provide effective control of hyperprolactinemia [70]. These findings emphasize that the problem is linked to the pathophysiology of uremia rather than to the dialysis modality.
The management of hyperprolactinemia in ESRD requires a selective, tailored approach. In clinical practice, serum PRL should be measured in dialysis patients presenting with otherwise unexplained sexual dysfunction, hypogonadism (e.g. low libido, erectile dysfunction, menstrual disturbances), galactorrhea, or infertility, and whenever gonadotropin or sex steroid abnormalities raise suspicion of a central dopaminergic disorder rather than isolated primary gonadal failure. Not all patients require treatment: in asymptomatic cases, a watchful waiting strategy may be adopted [71]. Pharmacological therapy is indicated in presence of significant clinical symptoms or when a concomitant prolactinoma is suspected. Dopamine agonists, such as cabergoline and bromocriptine, are the agents of choice: they have proven effective in lowering PRL levels and improving clinical manifestations, without requiring major dose adjustments in ESRD patients [72]. However, their use must be carefully weighed, particularly in patients with cardiovascular comorbidities, given the risks of hypotension, arrhythmias, and, with chronic therapy, valvulopathies [73–75]. Kidney transplantation remains the only truly curative strategy, capable of normalizing PRL levels in most cases within a few months of restored renal function [76]. This further underscores that endocrine alterations in ESRD are not isolated entities but rather direct consequences of chronic renal dysfunction, reversible with organ recovery.
In summary, hyperprolactinemia in dialysis patients is a highly prevalent and clinically significant condition that worsens sexual and reproductive function, contributes to hypogonadism, and potentially increases cardiovascular risk. Its management should be individualized, with selective use of pharmacological therapy and the awareness that kidney transplantation represents the only definitive intervention. A structured diagnostic-therapeutic pathway should begin with the exclusion of secondary causes and medications (neuroleptics, metoclopramide, verapamil, opioids), and PRL measurement should be primarily reserved for dialysis patients with suggestive clinical features such as sexual dysfunction, hypogonadism, galactorrhea, or infertility, rather than used as a universal screening test; subsequently, fasting blood sampling should be repeated, macroprolactin assessed, and the presence of CKD considered as a well-recognized cause of mild to moderate hyperprolactinemia [77]. In ESRD patients, neither conventional nor more frequent dialysis normalizes PRL levels; thus, treatment is reserved for symptomatic cases (sexual dysfunction, hypogonadism, galactorrhea) or when prolactinoma is present. Dopamine agonists (cabergoline, bromocriptine) are effective and generally do not require dose adjustment, although long-term clinical and echocardiographic monitoring is advisable given cumulative cabergoline exposure [78]. Finally, observational studies have reported associations between PRL and cardiovascular risk in CKD, but the evidence remains heterogeneous; prospective cohorts with long-term follow-up have shown conflicting results, underscoring the need for targeted future studies in ESRD [79].
HPT axis
From a practical standpoint, the primary consideration is to distinguish non-thyroidal illness (NTI) from ESRD-related abnormalities: in dialysis patients, reverse triiodothyronine (rT3) tends to be normal or low, whereas FT3 is the parameter most frequently reduced and of greatest prognostic relevance [80]. Second, analytical artifacts affecting FT4 must be considered, particularly those induced by heparin (even at low doses). Through lipolysis and the increase of free fatty acids, heparin may cause spurious elevations in FT4; therefore, blood sampling should ideally be performed before heparin administration or on a non-dialysis day, and results should be interpreted with caution [81–83]. Finally, the so-called “low T3 syndrome” is associated with poor prognosis in dialysis patients and has been shown to be an independent predictor of mortality in both HD and PD cohorts [84, 85]. Thyroid function abnormalities are well documented in ESRD patients and include changes in TSH, FT3, and FT4 levels, as well as alterations in their clearance and distribution [86]. TSH, in particular, exhibits atypical behavior: the response to exogenous TRH is blunted, pulsatile secretion is reduced, and plasma clearance is slowed by up to 57% compared with healthy individuals [87]. Moreover, the circadian rhythm of TSH, normally characterized by a nocturnal peak in subjects with preserved renal function, tends to be flattened in dialysis patients, reflecting central dysregulation [88, 89]. Measurement of TF4 in ESRD patients poses interpretative challenges. Analog-based assays may underestimate true values due to hypoalbuminemia, accumulation of uremic toxins competing for binding sites, and pharmacological interferences [90]. Particularly relevant is the effect of heparin, routinely used during HD, which stimulates lipoprotein lipase and causes transient rise in free fatty acid. These displace thyroxine from its binding proteins, leading to a spurious increase in FT4 [91]. This phenomenon must be carefully considered to avoid misdiagnosis. FT3 is the hormone most frequently affected. The so-called “low T3 syndrome” is reported in 65–75% of dialysis patients and is attributed to reduced peripheral conversion of T4 to T3, resulting from altered activity of type I and II deiodinases [92]. Chronic inflammation, protein-calorie malnutrition, and peripheral thyroid hormone resistance further contribute to this condition [93]. Importantly, it should not be regarded as a mere epiphenomenon: several studies have demonstrated that low-T3 syndrome is associated with poor outcomes and is an independent predictor of both cardiovascular and all-cause mortality [94]. Another diagnostic clue is rT3. In NTI, typically observed in critically ill patients, rT3 tends to be elevated; in contrast, in ESRD patients, rT3 levels are generally normal or low [95, 96]. This distinction allows accurate differentiation between the two conditions and appropriate interpretation of thyroid hormone alterations in dialysis patients.
Hypothyroidism
Hypothyroidism, in both overt and subclinical forms, is significantly more frequent in patients with CKD compared with general population. According to data from the National Health and Nutrition Examination Survey (NHANES), its prevalence may reach high values in advanced stages of CKD, with a further increase in patients undergoing dialysis therapy [97]. This condition represents not only a marker of advanced disease but also an independent prognostic factor for cardiovascular events and mortality [98]. The causes are multifactorial: chronic iodine retention, due to reduced renal clearance, may induce a persistent Wolff-Chaikoff effect with suppression of thyroid hormone synthesis [99]; chronic metabolic acidosis, commonly observed in uremic patients, contributes to increased TSH levels and reduced FT3 and FT4 concentrations [100]; finally, deficiencies of essential micronutrients such as selenium and zinc impair deiodinase activity, thereby exacerbating thyroid dysfunction [101]. Clinically, hypothyroidism in ESRD is associated with worsening cardiovascular function, including reduced myocardial contractility, increased arterial stiffness, and greater predisposition to vascular calcifications [102]. In PD patients, even subclinical forms have been correlated with reduced left ventricular ejection fraction, suggesting that mild alterations may have significant consequences [103]. Levothyroxine replacement therapy is indicated in overt form, in accordance with guideline recommendations [104], with individualized targets and titration, with carefully considering drug interactions and the dialysis regimen. In subclinical hypothyroidism, however, management remains debated: observational studies have suggested that replacement therapy may reduce cardiovascular risk and slow CKD progression [105], but randomized trials are needed to confirm these findings. The KDIGO guidelines recommend an individualized approach based on the patients’ risk profile, generally supporting treatment of subclinical hypothyroidism in CKD/ESRD when TSH is persistently and clearly elevated, in the presence of compatible symptoms, heart failure or high cardiovascular risk, while a watchful waiting strategy may be reasonable in asymptomatic patients with only mild TSH elevation [106].
Hyperthyroidism
Hyperthyroidism in patients with ESRD is rare, with a prevalence comparable to that of the general population [107]. Nevertheless, when present, it may have clinically relevant consequences. One of the main causes of hyperthyroidism in dialysis patients is iodine exposure from medications or contrast agents, which can trigger the Jod-Basedow phenomenon [108]. From a pathophysiological standpoint hyperthyroidism leads to an increase in glomerular filtration rate (GFR) of up to 18–25% [109], which may temporarily mask the severity of renal dysfunction but, at the same time, promotes the development of proteinuria and glomerulosclerosis [110]. In addition, it is associated with worsening arterial hypertension and accelerated progression of CKD [111].
Pharmacological treatment with methimazole does not require dose adjustment in CKD patients; however, up to 30–40% of the drug can be removed during a single HD session, and therefore administration is recommended immediately after dialysis [112]. In selected cases, treatment with radioiodine (I131) may be indicated, but its management in dialysis patients requires particular caution: isotope elimination occurs primarily through dialysis, and administration must be carefully planned in a multidisciplinary setting, ideally scheduled immediately after a dialysis session with a minimum interval of 12 h before the next one [113]. In patients with hyperparathyroidism or thyroid cancer who are candidates for I131 management, multidisciplinary coordinated planning is required: administration ideally immediately after an HD session, an appropriate interval before the next session, and strategies to maximize thyroid uptake while minimizing systemic exposure. In addition, special attention must be paid to the handling of spent dialysate and extracorporeal circuit components, which should be managed as radioactive waste according to local radiation-safety protocols, with appropriate shielding, storage, and disposal procedures to limit exposure of staff and the environment. The literature supports personalized approaches and dedicated protocols in dialysis patients [114].
Thyroid nodules and carcinoma
The overall prevalence of thyroid carcinoma in patients with CKD is comparable to that in the general population; however, some studies have reported a higher incidence and a more aggressive clinical course of papillary thyroid carcinoma in dialysis patients [115]. The causes of this association are not fully understood but may be related to chronic iodine exposure—due to reduced renal iodine clearance combined with repeated exposure to iodine-containing contrast media, antiseptic solutions, and some medications—as well as to the persistent inflammatory milieu and immune system alterations typical of uremia [116]. From a management perspective, the diagnosis and treatment of thyroid carcinoma in ESRD patients pose unique challenges. In particular, the use of radioiodine is complicated by reduced renal clearance and the need for its elimination through dialysis. This entails prolonged exposure risks not only for the patient but also for healthcare personnel [117]. For this reason, optimal management requires a multidisciplinary approach involving nephrologists, endocrinologists, oncologists, and nuclear medicine specialists.
Other endocrine axes
Beyond gonadal, thyroid, and PRL-related alterations, patients with ESRD exhibit dysfunctions involving other endocrine axes, which although less extensively studied, are clinically relevant [118]. In ESRD patients, basal cortisol secretion is generally preserved; however, several studies have demonstrated an attenuated response to stress stimuli such as severe infections, surgery, or hemodynamic shock [119]. This phenomenon, known as “relative adrenal insufficiency” appears to be related both to altered bioavailability of free cortisol—due to hypoalbuminemia and changes in transcortin—and to tissue resistance to glucocorticoids [120]. Clinically, this condition may manifest as an inadequate stress response, contributing to worse outcomes in critical situations.
One of the most severely affected axes in uremic patients is the vitamin D-PTH axis. Reduced activity of renal 1α-hydroxylase results in a marked decrease in calcitriol biosynthesis, leading to hypocalcemia and stimulation of PTH secretion [121]. The consequence is secondary hyperparathyroidism, which in turn promotes bone resorption and contributes to renal osteodystrophy. This alteration extends beyond bone metabolism. Deficiency of active vitamin D and excess PTH have been associated with sarcopenia, reduced muscular strength, impaired immune function, and increased susceptibility to infections [122]. In addition, elevated PTH contributes to vascular remodeling and arterial calcification, further exacerbating the already high cardiovascular risk in ESRD patients [123]. Overall, these alterations underscore how ESRD represents a systemic endocrine syndrome, in which impaired renal function triggers a cascade of hormonal dysfunctions with multisystem consequences. Integrated management of these axes therefore constitutes a crucial component of the comprehensive care of dialysis patients [124].
| Differences Between HD and PD on Endocrine Axes | ▴Top |
Dialysis modalities differ not only in technique and impact on quality of life but also in their consequences on the endocrine profile of patients. Although both PD and HD share the goal of replacing renal function, their effects on hormonal axes are not overlapping [125]. Several studies have demonstrated that male patients on HD exhibit lower mean testosterone levels compared with those on PD [126]. This difference may in part be explained by the greater hemodynamic instability of intermittent HD, metabolic fluctuations, and higher exposure to acute inflammatory states compared with PD, which provides more continuous and stable clearance, but it may also reflect baseline differences in case-mix, as PD patients are often younger, less comorbid, and more functionally independent. In women, sexual function appears to be less impaired in CAPD, with slightly higher FSFI scores than in HD patients [127]. This difference may reflect not only biological factors and the greater sense of autonomy and control associated with PD, which positively influence perceived quality of life and relational well-being, but also the fact that PD cohorts frequently include younger and overall healthier patients who are able and willing to perform dialysis themselves.
Hyperprolactinemia occurs in both dialysis modalities, but some studies have reported higher mean concentrations in HD patients, likely due to acute fluctuations in plasma PRL levels induced by dialysis sessions. In PD, by contrast, continuous clearance appears to provide greater stability, although absolute values are not normalized [128]. Routine heparin use in HD represents an additional factor capable of interfering with FT4 assays, complicating their clinical interpretation [129]. This issue is not present in PD patients, in whom thyroid hormone measurements are generally more stable and reliable. Patient-reported quality of life is generally better in PD, particularly with regard to sexual and relational domains [130]. This outcome may be attributed both to clinical factors (metabolic stability) and to psychosocial factors (autonomy, reduced hospitalization). Nevertheless, the choice of dialysis modality must always be individualized, taking into account comorbidities, family support, and personal preferences. These baseline differences in age, comorbidity burden, and functional status should be kept in mind when interpreting endocrine and sexual function differences between HD and PD populations.
| Clinical Management of Endocrine Alterations in ESRD | ▴Top |
The management of endocrine alterations in patients with ESRD represents a highly complex clinical challenge requiring an integrated, multidisciplinary, and patient-centered approach. Therapeutic strategies are based on three main pillars: systematic screening, individualized treatment, and multidisciplinary integration [131, 132]. One of the main limitations in routine clinical practice is the limited attention paid to endocrine screening in dialysis patients. Despite the well-documented prognostic role of parameters such as testosterone, FT3, and PRL [133], these assessments are not routinely included in follow-up protocols. A more proactive approach should include periodic measurement of total testosterone (and free testosterone in uncertain cases), estradiol, PRL, TSH, FT3, FT4, and active vitamin D [134]. In addition to hormonal assays, validated tools such as the (IIEF-5 for men and the FSFI for women are valuable instruments for assessing sexual function and relational well-being [135]. Incorporating these tools into nephrology clinics would allow early identification of at-risk patients, enabling timely intervention.
The treatment of endocrine alterations in ESRD cannot be standardized but must be tailored to the clinical profile of each patient.
Male hypogonadism
PDE-5i are first-line therapy for erectile dysfunction and have proven effective even in dialysis patients [136]. TRT may improve systemic symptoms and quality of life but should be reserved for selected cases, with close monitoring for risk of polycythemia, fluid retention, and cardiovascular complications [137]. In patients wishing to preserve fertility, alternatives such as clomiphene citrate and human gonadotropins offer safe options [138].
Female hypogonadism
Management relies on local estrogen therapy for genitourinary symptoms and, in selected cases, systemic hormone replacement therapy, preferably via the transdermal route, with careful evaluation of thromboembolic and cardiovascular risk [139].
Hyperprolactinemia
Does not always require treatment. In symptomatic cases or when a prolactinoma is present, dopamine agonists (cabergoline, bromocriptine) are the therapy of choice [140]. Their use must be closely monitored for potential cardiovascular adverse effects.
Hypothyroidism
Overt forms require levothyroxine replacement therapy. In subclinical hypothyroidism, management should be individualized, considering cardiovascular risk, age, and symptoms [141].
Hyperthyroidism
Management includes antithyroid drugs and, in selected cases, radioiodine therapy, with integrated scheduling of dialysis sessions [142].
Secondary hyperparathyroidism
Treatment includes active vitamin D (calcitriol and analogs) and Calcimimetics, in accordance with KDIGO guidelines [143].
Kidney transplantation represents the only truly curative strategy for most endocrine alterations in ESRD. After transplantation, testosterone, PRL, and thyroid hormone levels tend to normalize progressively [144]. However, long-term immunosuppressive therapy introduces new endocrine challenges, such as iatrogenic hypercortisolism, calcineurin inhibitor-induced metabolic syndrome, and post-transplant osteoporosis [145]. Continuous endocrinological management is therefore required even after restoration of renal function. The complexity of endocrine alterations in ESRD necessitates management involving multiple specialists: nephrologists, endocrinologists, andrologists, gynecologists, cardiologists, and dietitians. The creation of an integrated nephron-endocrinology pathway represents a concrete and desirable prospect for the future [146].
| Clinical Perspectives/Future Directions | ▴Top |
Endocrine alterations in patients with ESRD remain a largely understudied area, despite representing a significant determinant of quality of life, morbidity, and mortality. Their clinical implications range from bone metabolism to cardiovascular risk, from fertility to metabolic complications, outlining a scenario that requires systematic and integrated attention [147]. From a clinical care perspective, the integration of routine hormonal screening into follow-up protocols for dialysis patients should be prioritized. This should include periodic assessment of testosterone, estradiol, PRL, TSH, FT3, FT4, vitamin D, and PTH as well as standardized clinical tools for evaluating sexual function and quality of life [148]. A proactive approach would allow early identification of potentially treatable abnormalities and guide patients toward personalized therapeutic pathways. Therapeutic strategies should evolve toward greater personalization, taking into account both the clinical heterogeneity of ESRD patients and the complexity of pharmacological interactions. Randomized clinical trials are still needed to clarify the efficacy and safety of TRT, the management of subclinical hypothyroidism, and the use of dopamine agonists for hyperprolactinemia [149]. Furthermore, the impact of vitamin D supplementation strategies and drugs modulating mineral-bone metabolism (Calcimimetics) deserves prospective evaluation [150].
Despite a growing body of literature, several knowledge gaps remain. The precise molecular mechanisms through which anemia interferes with hypothalamic-pituitary axes are yet to be fully elucidated, as is the long-term relationship between endocrine dysfunction and cardiovascular outcomes [151]. Translational research may also help identify novel hormonal biomarkers predictive of prognosis and therapeutic response [152]. From an organizational perspective, it is essential to promote multidisciplinary collaboration models between nephrologists and endocrinologists, in order to overcome the current fragmentation of care. Integrated care pathways would optimize the management of endocrine complications, improve quality of life, and reduce mortality [153]. At the same time, the adoption of more physiological dialysis technique and the expansion of access to kidney transplantation remain key objectives to mitigate the endocrine impact of ESRD [154].
| Conclusions | ▴Top |
Endocrine disorders in ESRD remain an overlooked aspect of patient care, despite their major clinical impact. Hypogonadism, hyperprolactinemia, and thyroid dysfunction are not minor laboratory abnormalities but prognostic marker that influence survival and quality of life. Evidence links low testosterone and FT3 with increased mortality, underscoring the importance of systematic monitoring. Dialysis modality also matters: differences between HD and PD indicate that treatment choice should account for endocrine consequences as well as clinical and logistical factors. Optimal management requires regular screening, personalized treatment, and a multidisciplinary approach. Kidney transplantation is the only curative option, yet long-term endocrinological follow-up remains essential to manage complications for immunosuppression. Prospective studies and randomized trials are urgently needed to define the role of hormone replacement therapies in this vulnerable population, which is still underrepresented in the literature. Greater collaboration between nephrologists and endocrinologists is key to improving outcomes and quality of life.
Acknowledgments
None to declare.
Financial Disclosure
No funding was received for conducting this study.
Conflict of Interest
The authors declare no competing interests relevant to the contents of this article.
Informed Consent
Not applicable.
Author Contributions
Vincenzo Barbera: conceptualization, data interpretation, and critical revision of the manuscript for important intellectual content. Lorenzo D’Elia: contribution to the discussion and future perspectives sections; manuscript review. Luca Di Lullo: conceptual input and critical revision of the manuscript for important intellectual content. Paola Peverini: literature analysis and manuscript editing. Aldo Franculli: conceptualization, literature review, drafting of the manuscript, and coordination of the work. Pasquale Saporito: literature review and contribution to manuscript drafting. Andrea Dello Strologo: study supervision, critical revision, and final approval of the manuscript. Deborah Di Vico: data collection and literature analysis. Giovanni Otranto: data management and assistance in manuscript preparation. Annalisa Villani: contribution to data organization and editing of selected sections. Antonio Bellasi: scientific supervision, conceptual guidance, and critical revision for intellectual content. All authors read and approved the final version of the manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Mahmoud T, Borgi L. The interplay between nutrition, metabolic, and endocrine disorders in chronic kidney disease. Semin Nephrol. 2021;41(2):180-188.
doi pubmed - Leavey SF, Weitzel WF. Endocrine abnormalities in chronic renal failure. Endocrinol Metab Clin North Am. 2002;31(1):107-119.
doi pubmed - American Diabetes Association Professional Practice Committee for Diabetes. 11. Chronic kidney disease and risk management: standards of care in diabetes-2026. Diabetes Care. 2026;49(Supplement_1):S246-S260.
doi pubmed - Dembowska E, Jaron A, Gabrysz-Trybek E, Bladowska J, Gacek S, Trybek G. Quality of life in patients with end-stage renal disease undergoing hemodialysis. J Clin Med. 2022;11(6):1584.
doi pubmed - Carrero JJ, Stenvinkel P. Inflammation in end-stage renal disease—what have we learned in 10 years? Semin Dial. 2010;23(5):498-509.
doi pubmed - Stenvinkel P, Carrero JJ, Axelsson J, Lindholm B, Heimburger O, Massy Z. Emerging biomarkers for evaluating cardiovascular risk in the chronic kidney disease patient: how do new pieces fit into the uremic puzzle? Clin J Am Soc Nephrol. 2008;3(2):505-521.
doi pubmed - Carrero JJ, Qureshi AR, Parini P, Arver S, Lindholm B, Barany P, Heimburger O, et al. Low serum testosterone increases mortality risk among male dialysis patients. J Am Soc Nephrol. 2009;20(3):613-620.
doi pubmed - Ketteler M, Evenepoel P, Holden RM, Isakova T, Jorgensen HS, Komaba H, Nickolas TL, et al. Chronic kidney disease-mineral and bone disorder: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference. Kidney Int. 2025;107(3):405-423.
doi pubmed - Holley JL. The hypothalamic-pituitary axis in men and women with chronic kidney disease. Adv Chronic Kidney Dis. 2004;11(4):337-341.
pubmed - Cobo G, Lindholm B, Stenvinkel P. Chronic inflammation in end-stage renal disease and dialysis. Nephrol Dial Transplant. 2018;33(suppl_3):iii35-iii40.
doi pubmed - Castelo-Branco C, Cancelo MJ, Villero J, Nohales F, Julia MD. Management of post-menopausal vaginal atrophy and atrophic vaginitis. Maturitas. 2005;52(Suppl 1):S46-52.
doi pubmed - Collaborative Depression and Sexual Dysfunction in Hemodialysis Working Group, Vecchio M, Palmer S, De Berardis G, Craig J, Johnson D, et al. Prevalence and correlates of erectile dysfunction in men on chronic haemodialysis: a multinational cross-sectional study. Nephrol Dial Transplant. 2012;27(6):2479-2488.
doi pubmed - Chiang JM, Kaysen GA, Segal M, Chertow GM, Delgado C, Johansen KL. Low testosterone is associated with frailty, muscle wasting and physical dysfunction among men receiving hemodialysis: a longitudinal analysis. Nephrol Dial Transplant. 2019;34(5):802-810.
doi pubmed - Carrero JJ, Stenvinkel P. The vulnerable man: impact of testosterone deficiency on the uraemic phenotype. Nephrol Dial Transplant. 2012;27(11):4030-4041.
doi pubmed - Carrero JJ, Barany P, Yilmaz MI, Qureshi AR, Sonmez A, Heimburger O, Ozgurtas T, et al. Testosterone deficiency is a cause of anaemia and reduced responsiveness to erythropoiesis-stimulating agents in men with chronic kidney disease. Nephrol Dial Transplant. 2012;27(2):709-715.
doi pubmed - Rytz CL, Kochaksaraei GS, Skeith L, Ronksley PE, Dumanski SM, Robert M, Ahmed SB. Menstrual abnormalities and reproductive lifespan in females with CKD: a systematic review and meta-analysis. Clin J Am Soc Nephrol. 2022;17(12):1742-1753.
doi pubmed - Ahmed SB, Ramesh S. Sex hormones in women with kidney disease. Nephrol Dial Transplant. 2016;31(11):1787-1795.
doi pubmed - Xu H, Brusselaers N, Lindholm B, Zoccali C, Carrero JJ. Thyroid function test derangements and mortality in dialysis patients: a systematic review and meta-analysis. Am J Kidney Dis. 2016;68(6):923-932.
doi pubmed - Ho YF, Li IC. The influence of different dialysis modalities on the quality of life of patients with end-stage renal disease: A systematic literature review. Psychol Health. 2016;31(12):1435-1465.
doi pubmed - Gungor O, Kocyigit I, Carrero JJ, Yilmaz MI. Hormonal changes in hemodialysis patients: Novel risk factors for mortality? Semin Dial. 2017;30(5):446-452.
doi pubmed - Bilha SC, Hogas S, Hogas M, Marcu S, Leustean L, Ungureanu MC, Branisteanu DD, et al. Thyroid, gonadal and adrenal dysfunction in kidney transplant recipients: a review for the clinician. Biomolecules. 2023;13(6):920.
doi pubmed - Mulhall JP, Trost LW, Brannigan RE, Kurtz EG, Redmon JB, Chiles KA, Lightner DJ, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200(2):423-432.
doi pubmed - Strippoli GF, Collaborative D, Sexual Dysfunction in Hemodialysis Working G, Vecchio M, Palmer S, De Berardis G, Craig J, et al. Sexual dysfunction in women with ESRD requiring hemodialysis. Clin J Am Soc Nephrol. 2012;7(6):974-981.
doi pubmed - Iglesias P, Carrero JJ, Diez JJ. Gonadal dysfunction in men with chronic kidney disease: clinical features, prognostic implications and therapeutic options. J Nephrol. 2012;25(1):31-42.
doi pubmed - Chang DH, Dumanski SM, Ahmed SB. Female reproductive and gynecologic considerations in chronic kidney disease: adolescence and young adulthood. Kidney Int Rep. 2022;7(2):152-164.
doi pubmed - Daschner M, Philippin B, Nguyen T, Wiesner RJ, Walz C, Oh J, Sandow J, et al. Circulating inhibitor of gonadotropin releasing hormone secretion by hypothalamic neurons in uremia. Kidney Int. 2002;62(5):1582-1590.
doi pubmed - Holdsworth S, Atkins RC, de Kretser DM. The pituitary-testicular axis in men with chronic renal failure. N Engl J Med. 1977;296(22):1245-1249.
doi pubmed - Schaefer F, Vogel M, Kerkhoff G, Woitzik J, Daschner M, Mehls O. Experimental uremia affects hypothalamic amino acid neurotransmitter milieu. J Am Soc Nephrol. 2001;12(6):1218-1227.
doi pubmed - Huang W, Molitch ME. Prolactin and other pituitary disorders in kidney disease. Semin Nephrol. 2021;41(2):156-167.
doi pubmed - Buvat J. Hyperprolactinemia and sexual function in men: a short review. Int J Impot Res. 2003;15(5):373-377.
doi pubmed - Wang Y, Chen L, Xie L, Li L, Li X, Li H, Liu J, et al. Interleukin 6 inhibits the differentiation of rat stem Leydig cells. Mol Cell Endocrinol. 2018;472:26-39.
doi pubmed - Stenvinkel P, Ketteler M, Johnson RJ, Lindholm B, Pecoits-Filho R, Riella M, Heimburger O, et al. IL-10, IL-6, and TNF-alpha: central factors in the altered cytokine network of uremia—the good, the bad, and the ugly. Kidney Int. 2005;67(4):1216-1233.
doi pubmed - He Z, Yin G, Li QQ, Zeng Q, Duan J. Diabetes mellitus causes male reproductive dysfunction: a review of the evidence and mechanisms. In Vivo. 2021;35(5):2503-2511.
doi pubmed - Genchi VA, Rossi E, Lauriola C, D'Oria R, Palma G, Borrelli A, Caccioppoli C, et al. Adipose tissue dysfunction and obesity-related male hypogonadism. Int J Mol Sci. 2022;23(15):8194.
doi pubmed - Romejko K, Rymarz A, Sadownik H, Niemczyk S. Testosterone deficiency as one of the major endocrine disorders in chronic kidney disease. Nutrients. 2022;14(16):3438.
doi pubmed - Yu J, Ravel VA, You AS, Streja E, Rivara MB, Potukuchi PK, Brunelli SM, et al. Association between testosterone and mortality risk among U.S. males receiving dialysis. Am J Nephrol. 2017;46(3):195-203.
doi pubmed - Carrero JJ, Qureshi AR, Nakashima A, Arver S, Parini P, Lindholm B, Barany P, et al. Prevalence and clinical implications of testosterone deficiency in men with end-stage renal disease. Nephrol Dial Transplant. 2011;26(1):184-190.
doi pubmed - Matsumoto AM. Diagnosis and evaluation of hypogonadism. Endocrinol Metab Clin North Am. 2022;51(1):47-62.
doi pubmed - De Silva NL, Papanikolaou N, Grossmann M, Antonio L, Quinton R, Anawalt BD, Jayasena CN. Male hypogonadism: pathogenesis, diagnosis, and management. Lancet Diabetes Endocrinol. 2024;12(10):761-774.
doi pubmed - Bao Y, Johansen KL. Diagnosis and treatment of low testosterone among patients with end-stage renal disease. Semin Dial. 2015;28(3):259-265.
doi pubmed - Skiba R, Rymarz A, Matyjek A, Dymus J, Wozniak-Kosek A, Syrylo T, Zielinski H, et al. Testosterone replacement therapy in chronic kidney disease patients. Nutrients. 2022;14(16):3444.
doi pubmed - Wu YC, Sung WW. Clomiphene citrate treatment as an alternative therapeutic approach for male hypogonadism: mechanisms and clinical implications. Pharmaceuticals (Basel). 2024;17(9):1233.
doi pubmed - Schlegel PN, Sigman M, Collura B, De Jonge CJ, Eisenberg ML, Lamb DJ, Mulhall JP, et al. Diagnosis and treatment of infertility in men: AUA/ASRM guideline part II. Fertil Steril. 2021;115(1):62-69.
doi pubmed - Turk S, Solak Y, Kan S, Atalay H, Kilinc M, Agca E, Bodur S. Effects of sildenafil and vardenafil on erectile dysfunction and health-related quality of life in haemodialysis patients: a prospective randomized crossover study. Nephrol Dial Transplant. 2010;25(11):3729-3733.
doi pubmed - Lasaponara F, Sedigh O, Pasquale G, Bosio A, Rolle L, Ceruti C, Timpano M, et al. Phosphodiesterase type 5 inhibitor treatment for erectile dysfunction in patients with end-stage renal disease receiving dialysis or after renal transplantation. J Sex Med. 2013;10(11):2798-2814.
doi pubmed - Heidelbaugh JJ, Belakovskiy A. Testosterone replacement therapy for male hypogonadism. Am Fam Physician. 2024;109(6):543-549.
pubmed - Deebel NA, Matthew AN, Loloi J, Bernstein AP, Thirumavalavan N, Ramasamy R. Testosterone deficiency in men with end stage renal disease and kidney transplantation: a narrative review. Int J Impot Res. 2025;37(4):271-277.
doi pubmed - Zhang K, Zhao L, Bin Y, Guo M, Zhou X, Li M, Han L, et al. Serum prolactin and gonadal hormones in hemodialysis women: a meta-analysis. BMC Endocr Disord. 2023;23(1):203.
doi pubmed - Pyrgidis N, Mykoniatis I, Tishukov M, Sokolakis I, Nigdelis MP, Sountoulides P, Hatzichristodoulou G, et al. Sexual dysfunction in women with end-stage renal disease: a systematic review and meta-analysis. J Sex Med. 2021;18(5):936-945.
doi pubmed - Zeighami S, Dehghankhalili S, Heiran K, Azarchehry SP, Heiran A, Sayadi M, Azadian F. Comparison of male and female sexual dysfunction between hemodialysis and peritoneal dialysis in patients with end-stage renal disease: an analytical cross-sectional study. Int J Endocrinol. 2022;2022:9404025.
doi pubmed - Kettas E, Cayan F, Akbay E, Kiykim A, Cayan S. Sexual dysfunction and associated risk factors in women with end-stage renal disease. J Sex Med. 2008;5(4):872-877.
doi pubmed - Juergensen E, Wuerth D, Finkelstein SH, Juergensen PH, Bekui A, Finkelstein FO. Hemodialysis and peritoneal dialysis: patients' assessment of their satisfaction with therapy and the impact of the therapy on their lives. Clin J Am Soc Nephrol. 2006;1(6):1191-1196.
doi pubmed - Davenport A. Frailty, appendicular lean mass, osteoporosis and osteosarcopenia in peritoneal dialysis patients. J Nephrol. 2022;35(9):2333-2340.
doi pubmed - Xiang T, Fu P, Zhou L. Sarcopenia and osteosarcopenia among patients undergoing hemodialysis. Front Endocrinol (Lausanne). 2023;14:1181139.
doi pubmed - Pinkerton JV. Hormone therapy for postmenopausal women. N Engl J Med. 2020;382(5):446-455.
doi pubmed - Cevik EC, Erel CT, Ozcivit Erkan IB, Sarafidis P, Armeni E, Fistonic I, Hillard T, et al. Chronic kidney disease and menopausal health: An EMAS clinical guide. Maturitas. 2025;192:108145.
doi pubmed - Luo L, Xiao C, Xiang Q, Zhu Z, Liu Y, Wang J, Deng Y, et al. Significant increase of sexual dysfunction in patients with renal failure receiving renal replacement therapy: a systematic review and meta-analysis. J Sex Med. 2020;17(12):2382-2393.
doi pubmed - Flores VA, Pal L, Manson JE. Hormone therapy in menopause: concepts, controversies, and approach to treatment. Endocr Rev. 2021;42(6):720-752.
doi pubmed - Arnautu AM, Nimigean VR, Nacea-Radu CA, Tilici DM, Paun DL. Menopausal hormone therapy-risks, benefits and emerging options: a narrative review. Int J Mol Sci. 2025;26(22):11098.
doi pubmed - Rojhani E, Rahmati M, Firouzi F, Ziaeefar P, Soudmand SA, Azizi F, Tehrani FR, et al. Prolactin levels and chronic kidney disease and the subsequent risk of cardiovascular events: A long term population based cohort study. Sci Rep. 2025;15(1):7198.
doi pubmed - Cowden EA, Ratcliffe WA, Ratcliffe JG, Dobbie JW, Kennedy AC. Hyperprolactinaemia in renal disease. Clin Endocrinol (Oxf). 1978;9(3):241-248.
doi pubmed - Ramirez G, O'Neill WM, Jr., Bloomer HA, Jubiz W. Abnormalities in the regulation of prolactin in patients with chronic renal failure. J Clin Endocrinol Metab. 1977;45(4):658-661.
doi pubmed - Ben-Jonathan N, Hnasko R. Dopamine as a prolactin (PRL) inhibitor. Endocr Rev. 2001;22(6):724-763.
doi pubmed - Rusu CC, Moldovan D, Potra A, Tirinescu D, Ticala M, Maslyennikov Y, Barar A, et al. The interplay of prolactin with inflammatory nutritional markers and NT-proBNP in chronic kidney disease. Int J Mol Sci. 2025;26(13):6347.
doi pubmed - Haidenberg-David F, Sidauy-Adissi J, Moscona-Nissan A, Jonguitud-Zumaya E, Fugarolas-Morinelli M, Martinez-Mendoza F, Taniguchi-Ponciano K, et al. Overview of hyperprolactinemia: general approach and reproductive health implications. Arch Med Res. 2024;55(8):103102.
doi pubmed - De Rosa M, Zarrilli S, Di Sarno A, Milano N, Gaccione M, Boggia B, Lombardi G, et al. Hyperprolactinemia in men: clinical and biochemical features and response to treatment. Endocrine. 2003;20(1-2):75-82.
doi pubmed - Bhasin S, Enzlin P, Coviello A, Basson R. Sexual dysfunction in men and women with endocrine disorders. Lancet. 2007;369(9561):597-611.
doi pubmed - Dourado M, Cavalcanti F, Vilar L, Cantilino A. Relationship between prolactin, chronic kidney disease, and cardiovascular risk. Int J Endocrinol. 2020;2020:9524839.
doi pubmed - Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, Wass JA, et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-288.
doi pubmed - van Eps C, Hawley C, Jeffries J, Johnson DW, Campbell S, Isbel N, Mudge DW, et al. Changes in serum prolactin, sex hormones and thyroid function with alternate nightly nocturnal home haemodialysis. Nephrology (Carlton). 2012;17(1):42-47.
doi pubmed - Wang AT, Mullan RJ, Lane MA, Hazem A, Prasad C, Gathaiya NW, Fernandez-Balsells MM, et al. Treatment of hyperprolactinemia: a systematic review and meta-analysis. Syst Rev. 2012;1:33.
doi pubmed - Auriemma RS, Pirchio R, Pivonello C, Garifalos F, Colao A, Pivonello R. Approach to the patient with prolactinoma. J Clin Endocrinol Metab. 2023;108(9):2400-2423.
doi pubmed - Valassi E, Klibanski A, Biller BM. Clinical Review#: Potential cardiac valve effects of dopamine agonists in hyperprolactinemia. J Clin Endocrinol Metab. 2010;95(3):1025-1033.
doi pubmed - Khare S, Lila AR, Patil R, Phadke M, Kerkar P, Bandgar T, Shah NS. Long-term cardiac (valvulopathy) safety of cabergoline in prolactinoma. Indian J Endocrinol Metab. 2017;21(1):154-159.
doi pubmed - Samson SL, Ezzat S. AACE/ACE disease state clinical review: dopamine agonists for hyperprolactinemia and the risk of cardiac valve disease. Endocr Pract. 2014;20(6):608-616.
doi pubmed - Saha MT, Saha HH, Niskanen LK, Salmela KT, Pasternack AI. Time course of serum prolactin and sex hormones following successful renal transplantation. Nephron. 2002;92(3):735-737.
doi pubmed - Practice Committee of the American Society for Reproductive Medicine. Current evaluation of amenorrhea: a committee opinion. Fertil Steril. 2024;122(1):52-61.
doi pubmed - Fleseriu M, Varlamov EV, Akirov A, Langlois F, Petersenn S, Melmed S. Prolactin-secreting adenomas: pathogenesis, diagnosis, and management. Lancet Diabetes Endocrinol. 2025;13(10):874-890.
doi pubmed - Georgiopoulos GA, Stamatelopoulos KS, Lambrinoudaki I, Lykka M, Kyrkou K, Rizos D, Creatsa M, et al. Prolactin and preclinical atherosclerosis in menopausal women with cardiovascular risk factors. Hypertension. 2009;54(1):98-105.
doi pubmed - Lim VS. Thyroid function in patients with chronic renal failure. Am J Kidney Dis. 2001;38(4 Suppl 1):S80-84.
doi pubmed - Jansen HI, van Herwaarden AE, Huijgen HJ, Vervloet MG, Hillebrand JJ, Boelen A, Heijboer AC. Lower accuracy of testosterone, cortisol, and free T4 measurements using automated immunoassays in people undergoing hemodialysis. Clin Chem Lab Med. 2023;61(8):1436-1445.
doi pubmed - Stevenson HP, Archbold GP, Johnston P, Young IS, Sheridan B. Misleading serum free thyroxine results during low molecular weight heparin treatment. Clin Chem. 1998;44(5):1002-1007.
pubmed - Nishikawa M, Ogawa Y, Yoshikawa N, Yoshimura M, Toyoda N, Shouzu A, Inada M. Plasma free thyroxine (FT4) concentrations during hemodialysis in patients with chronic renal failure: effects of plasma non-esterified fatty acids on FT4 measurement. Endocr J. 1996;43(5):487-493.
doi pubmed - Drechsler C, Schneider A, Gutjahr-Lengsfeld L, Kroiss M, Carrero JJ, Krane V, Allolio B, et al. Thyroid function, cardiovascular events, and mortality in diabetic hemodialysis patients. Am J Kidney Dis. 2014;63(6):988-996.
doi pubmed - Zoccali C, Mallamaci F, Tripepi G, Cutrupi S, Pizzini P. Low triiodothyronine and survival in end-stage renal disease. Kidney Int. 2006;70(3):523-528.
doi pubmed - Sanai T, Okamura K, Onoue T, Ono T, Motomura K, Miyazono M, Shimamatsu K. Hemodilution impacts assessment of thyroid status before and after hemodialysis in patients with end-stage renal disease. Am J Nephrol. 2021;51(12):988-994.
doi pubmed - Duntas L, Wolf CF, Keck FS, Rosenthal J. Thyrotropin-releasing hormone: pharmacokinetic and pharmacodynamic properties in chronic renal failure. Clin Nephrol. 1992;38(4):214-218.
pubmed - Soylu H, Ersoy R, Keske PB, Tuncay ME, Ates C, Cakir B, Yalcin B. The diurnal change of thyroid-stimulating hormone and the effect of this change on thyroid functions in patients with chronic kidney disease. Endocrine. 2023;82(3):580-585.
doi pubmed - Yonemura K, Nakajima T, Suzuki T, Ando S, Genma R, Nakamura H, Hishida A. Low free thyroxine concentrations and deficient nocturnal surge of thyroid-stimulating hormone in haemodialysed patients compared with undialysed patients. Nephrol Dial Transplant. 2000;15(5):668-672.
doi pubmed - d'Herbomez M, Forzy G, Gasser F, Massart C, Beaudonnet A, Sapin R. Clinical evaluation of nine free thyroxine assays: persistent problems in particular populations. Clin Chem Lab Med. 2003;41(7):942-947.
doi pubmed - Burch HB. Drug effects on the thyroid. N Engl J Med. 2019;381(8):749-761.
doi pubmed - Horacek J, Dusilova Sulkova S, Kubisova M, Safranek R, Malirova E, Kalousova M, Svilias I, et al. Thyroid hormone abnormalities in hemodialyzed patients: low triiodothyronine as well as high reverse triiodothyronine are associated with increased mortality. Physiol Res. 2012;61(5):495-501.
doi pubmed - Zoccali C, Tripepi G, Cutrupi S, Pizzini P, Mallamaci F. Low triiodothyronine: a new facet of inflammation in end-stage renal disease. J Am Soc Nephrol. 2005;16(9):2789-2795.
doi pubmed - Meuwese CL, Dekker FW, Lindholm B, Qureshi AR, Heimburger O, Barany P, Stenvinkel P, et al. Baseline levels and trimestral variation of triiodothyronine and thyroxine and their association with mortality in maintenance hemodialysis patients. Clin J Am Soc Nephrol. 2012;7(1):131-138.
doi pubmed - Peeters RP, Wouters PJ, van Toor H, Kaptein E, Visser TJ, Van den Berghe G. Serum 3,3',5'-triiodothyronine (rT3) and 3,5,3'-triiodothyronine/rT3 are prognostic markers in critically ill patients and are associated with postmortem tissue deiodinase activities. J Clin Endocrinol Metab. 2005;90(8):4559-4565.
doi pubmed - Langouche L, Lehmphul I, Perre SV, Kohrle J, Van den Berghe G. Circulating 3-T1AM and 3,5-T2 in critically ill patients: a cross-sectional observational study. Thyroid. 2016;26(12):1674-1680.
doi pubmed - Taylor PN, Medici MM, Hubalewska-Dydejczyk A, Boelaert K. Hypothyroidism. Lancet. 2024;404(10460):1347-1364.
doi pubmed - Koo HM, Kim CH, Doh FM, Lee MJ, Kim EJ, Han JH, Han JS, et al. The impact of low triiodothyronine levels on mortality is mediated by malnutrition and cardiac dysfunction in incident hemodialysis patients. Eur J Endocrinol. 2013;169(4):409-419.
doi pubmed - Kutlay S, Atli T, Koseogullari O, Nergizoglu G, Duman N, Gullu S. Thyroid disorders in hemodialysis patients in an iodine-deficient community. Artif Organs. 2005;29(4):329-332.
doi pubmed - Wiederkehr MR, Kalogiros J, Krapf R. Correction of metabolic acidosis improves thyroid and growth hormone axes in haemodialysis patients. Nephrol Dial Transplant. 2004;19(5):1190-1197.
doi pubmed - Kohrle J. Selenium, iodine and iron-essential trace elements for thyroid hormone synthesis and metabolism. Int J Mol Sci. 2023;24(4):3393.
doi pubmed - Razvi S, Jabbar A, Pingitore A, Danzi S, Biondi B, Klein I, Peeters R, et al. Thyroid Hormones and Cardiovascular Function and Diseases. J Am Coll Cardiol. 2018;71(16):1781-1796.
doi pubmed - Kang EW, Nam JY, Yoo TH, Shin SK, Kang SW, Han DS, Han SH. Clinical implications of subclinical hypothyroidism in continuous ambulatory peritoneal dialysis patients. Am J Nephrol. 2008;28(6):908-913.
doi pubmed - Jonklaas J, Bianco AC, Bauer AJ, Burman KD, Cappola AR, Celi FS, Cooper DS, et al. Guidelines for the treatment of hypothyroidism: prepared by the american thyroid association task force on thyroid hormone replacement. Thyroid. 2014;24(12):1670-1751.
doi pubmed - Yu OHY, Filliter C, Filion KB, Platt RW, Grad R, Rad MV, Renoux C. Levothyroxine treatment for subclinical hypothyroidism and risk of adverse renal outcomes: a population-based cohort study. CMAJ. 2025;197(26):E744-E753.
doi pubmed - Cheng Y, Hu H, Li W, Nie S, Zhou S, Chen Y, Cao T, et al. Chronic kidney disease and thyroid hormones. J Clin Endocrinol Metab. 2025;110(8):e2446-e2455.
doi pubmed - Wiersinga WM, Poppe KG, Effraimidis G. Hyperthyroidism: aetiology, pathogenesis, diagnosis, management, complications, and prognosis. Lancet Diabetes Endocrinol. 2023;11(4):282-298.
doi pubmed - Chaker L, Cooper DS, Walsh JP, Peeters RP. Hyperthyroidism. Lancet. 2024;403(10428):768-780.
doi pubmed - Mariani LH, Berns JS. The renal manifestations of thyroid disease. J Am Soc Nephrol. 2012;23(1):22-26.
doi pubmed - van Hoek I, Daminet S. Interactions between thyroid and kidney function in pathological conditions of these organ systems: a review. Gen Comp Endocrinol. 2009;160(3):205-215.
doi pubmed - Allam MM, El-Zawawy HT, El-Zawawy TH. Renal function changes in patients with subclinical hyperthyroidism: a novel postulated mechanism. Endocrine. 2023;82(1):78-86.
doi pubmed - Holst JP, Burman KD, Atkins F, Umans JG, Jonklaas J. Radioiodine therapy for thyroid cancer and hyperthyroidism in patients with end-stage renal disease on hemodialysis. Thyroid. 2005;15(12):1321-1331.
doi pubmed - AlSadi R, Aziz LC, Bohan M, Dewji S, Bouhali O, Djekidel M. Clinical management of end-stage renal disease patients on dialysis receiving radioactive iodine treatment. Clin Nucl Med. 2021;46(12):977-982.
doi pubmed - Lee SY, Pearce EN. Hyperthyroidism: a review. JAMA. 2023;330(15):1472-1483.
doi pubmed - Demirer AN, Kemal Y, Gursoy A, Sahin M, Tutuncu NB. Clinicopathological characteristics of thyroid cancer in patients on dialysis for end-stage renal disease. Thyroid. 2008;18(1):45-50.
doi pubmed - Malyszko J, Tesarova P, Capasso G, Capasso A. The link between kidney disease and cancer: complications and treatment. Lancet. 2020;396(10246):277-287.
doi pubmed - Vermandel M, Debruyne P, Beron A, Devos L, Talbot A, Legrand JF, Provot F, et al. Management of patients with renal failure undergoing dialysis during (131)I therapy for thyroid cancer. J Nucl Med. 2020;61(8):1161-1170.
doi pubmed - Feneberg R, Schaefer F, Veldhuis JD. Neuroendocrine adaptations in renal disease. Pediatr Nephrol. 2003;18(6):492-497.
doi pubmed - Sagmeister MS, Harper L, Hardy RS. Cortisol excess in chronic kidney disease - a review of changes and impact on mortality. Front Endocrinol (Lausanne). 2022;13:1075809.
doi pubmed - Teblick A, Peeters B, Langouche L, Van den Berghe G. Adrenal function and dysfunction in critically ill patients. Nat Rev Endocrinol. 2019;15(7):417-427.
doi pubmed - Reiss AB, Miyawaki N, Moon J, Kasselman LJ, Voloshyna I, D'Avino R, Jr., De Leon J. CKD, arterial calcification, atherosclerosis and bone health: Inter-relationships and controversies. Atherosclerosis. 2018;278:49-59.
doi pubmed - Gal-Moscovici A, Sprague SM. Role of vitamin D deficiency in chronic kidney disease. J Bone Miner Res. 2007;22(Suppl 2):V91-94.
doi pubmed - Duque EJ, Elias RM, Moyses RMA. Parathyroid hormone: a uremic toxin. Toxins (Basel). 2020;12(3):189.
doi pubmed - Vanholder R, Fouque D, Glorieux G, Heine GH, Kanbay M, Mallamaci F, Massy ZA, et al. Clinical management of the uraemic syndrome in chronic kidney disease. Lancet Diabetes Endocrinol. 2016;4(4):360-373.
doi pubmed - Tekkarismaz N, Tunel M, Ozer C. Dialysis modality and sexual dysfunction in male patients. Andrologia. 2020;52(10):e13735.
doi pubmed - Lesniak K, Rymarz A, Sobol M, Dymus J, Wozniak-Kosek A, Niemczyk S. Testosterone deficiency and nutritional parameters as predictors of all-cause mortality among male dialysis patients. Nutrients. 2022;14(21):4461.
doi pubmed - Guglielmi KE. Women and ESRD: modalities, survival, unique considerations. Adv Chronic Kidney Dis. 2013;20(5):411-418.
doi pubmed - Rasche FM, Rettig R, Frese T, Rasche WG, Barinka F, Roesl G, Keller F, et al. The pituitary-thyroid axis and prolactin secretion in hemodialysis patients in two endemic regions of Eastern Germany. Exp Clin Endocrinol Diabetes. 2018;126(6):349-356.
doi pubmed - Ita JP, Lidija O, Lori SN, Tea VM, Jelena S, Ivan B. Thyroid function trends in dialysis: unveiling peritoneal and hemodialysis disparities. Semin Dial. 2024;37(6):438-444.
doi pubmed - Sanai T, Nagashima A, Okamura K, Rikitake S, Fukuda M, Onozawa K, Sanematsu M, et al. Thyroid function in patients on continuous ambulatory peritoneal dialysis in comparison with chronic kidney disease. Clin Nephrol. 2018;89(3):181-186.
doi pubmed - Wouk N. End-stage renal disease: medical management. Am Fam Physician. 2021;104(5):493-499.
pubmed - Goodbred AJ, Langan RC. Chronic kidney disease: prevention, diagnosis, and treatment. Am Fam Physician. 2023;108(6):554-561.
pubmed - Lesniak K, Rymarz A, Sobol M, Niemczyk S. Low free triiodothyronine as a more sensitive predictor of survival than total testosterone among dialysis men. Nutrients. 2023;15(3):595.
doi pubmed - Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl (2011). 2017;7(1):1-59.
doi pubmed - Luu HH, Ryan J, Toussaint ND. A narrative review of routine haematological and biochemical parameter monitoring in maintenance haemodialysis patients and comparison of clinical guidelines. BMC Nephrol. 2026;27(1):19.
doi pubmed - Salonia A, Capogrosso P, Boeri L, Cocci A, Corona G, Dinkelman-Smit M, Falcone M, et al. European Association of Urology Guidelines on Male Sexual and Reproductive Health: 2025 update on male hypogonadism, erectile dysfunction, premature ejaculation, and Peyronie's disease. Eur Urol. 2025;88(1):76-102.
doi pubmed - Canguven O, Aykose G, Albayrak S, Goktas C, Horuz R, Yencilek F. Efficacy of testosterone gel in the treatment of erectile dysfunction in hypogonadal hemodialysis patients: a pilot study. Int J Impot Res. 2010;22(2):140-145.
doi pubmed - Isidori AM, Buvat J, Corona G, Goldstein I, Jannini EA, Lenzi A, Porst H, et al. A critical analysis of the role of testosterone in erectile function: from pathophysiology to treatment-a systematic review. Eur Urol. 2014;65(1):99-112.
doi pubmed - Palmer BF, Clegg DJ. Gonadal dysfunction in chronic kidney disease. Rev Endocr Metab Disord. 2017;18(1):117-130.
doi pubmed - Tritos NA, Miller KK. Diagnosis and management of pituitary adenomas: a review. JAMA. 2023;329(16):1386-1398.
doi pubmed - Rhee CM. Thyroid disease in end-stage renal disease. Curr Opin Nephrol Hypertens. 2019;28(6):621-630.
doi pubmed - Suzuki M, Yoshimura Noh J, Sugino K, Ito K. Outpatient (131)I treatment for a patient with Graves' disease receiving hemodialysis. Intern Med. 2016;55(22):3309-3313.
doi pubmed - Miedziaszczyk M, Idasiak-Piechocka I, Wisniewski OW, Lacka K. A systematic review of the pharmacotherapy of secondary hyperparathyroidism (SHPT) in grades 3-5 Chronic Kidney Disease (CKD). Eur Rev Med Pharmacol Sci. 2022;26(1):232-239.
doi pubmed - Eckersten D, Giwercman A, Pihlsgard M, Bruun L, Christensson A. Impact of kidney transplantation on reproductive hormone levels in males: a longitudinal study. Nephron. 2018;138(3):192-201.
doi pubmed - Gosmanova EO, Tangpricha V, Gosmanov AR. Endocrine-metabolic pathophysiologic conditions and treatment approaches after kidney transplantation. Endocr Pract. 2012;18(4):579-590.
doi pubmed - de Boer IH, Khunti K, Sadusky T, Tuttle KR, Neumiller JJ, Rhee CM, Rosas SE, et al. Diabetes management in chronic kidney disease: a consensus report by the American Diabetes Association (ADA) and Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2022;102(5):974-989.
doi pubmed - Covic A, Vervloet M, Massy ZA, Torres PU, Goldsmith D, Brandenburg V, Mazzaferro S, et al. Bone and mineral disorders in chronic kidney disease: implications for cardiovascular health and ageing in the general population. Lancet Diabetes Endocrinol. 2018;6(4):319-331.
doi pubmed - Kim DH, Cho A, Park HC, Kim BY, Lee M, Kim GO, Kim J, et al. Regular laboratory testing and patient survival among patients undergoing maintenance hemodialysis: a Korean nationwide cohort study. Sci Rep. 2023;13(1):18360.
doi pubmed - Kovesdy CP. Clinical trials in end-stage renal disease-priorities and challenges. Nephrol Dial Transplant. 2019;34(7):1084-1089.
doi pubmed - Jorgensen HS, Vervloet M, Cavalier E, Bacchetta J, de Borst MH, Bover J, Cozzolino M, et al. The role of nutritional vitamin D in chronic kidney disease-mineral and bone disorder in children and adults with chronic kidney disease, on dialysis, and after kidney transplantation-a European consensus statement. Nephrol Dial Transplant. 2025;40(4):797-822.
doi pubmed - Shoji T, Emoto M, Nishizawa Y, Inaba M. Endocrine and metabolic changes affecting cardiovascular disease in dialysis patients. J Ren Nutr. 2015;25(2):223-225.
doi pubmed - Silva NR, Picolo BU, de Sousa LCM, Dos Santos MS, Polveiro RC, Almeida-Souza HO, Martins MM, et al. Urine proteomics for detection of potential biomarkers for end-stage renal disease. Int J Mol Sci. 2025;26(12):5429.
doi pubmed - Pages H, Kosmadakis G, Kanouni FE, Necoara A, Baudenon J, Deville C, Enache I, et al. Evaluation of multidisciplinary care pathways on kidney function in patients with advanced chronic kidney disease: multicenter study. J Nephrol. 2025;38(9):2775-2784.
doi pubmed - Robinson BM, Akizawa T, Jager KJ, Kerr PG, Saran R, Pisoni RL. Factors affecting outcomes in patients reaching end-stage kidney disease worldwide: differences in access to renal replacement therapy, modality use, and haemodialysis practices. Lancet. 2016;388(10041):294-306.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
World Journal of Nephrology and Urology is published by Elmer Press Inc.