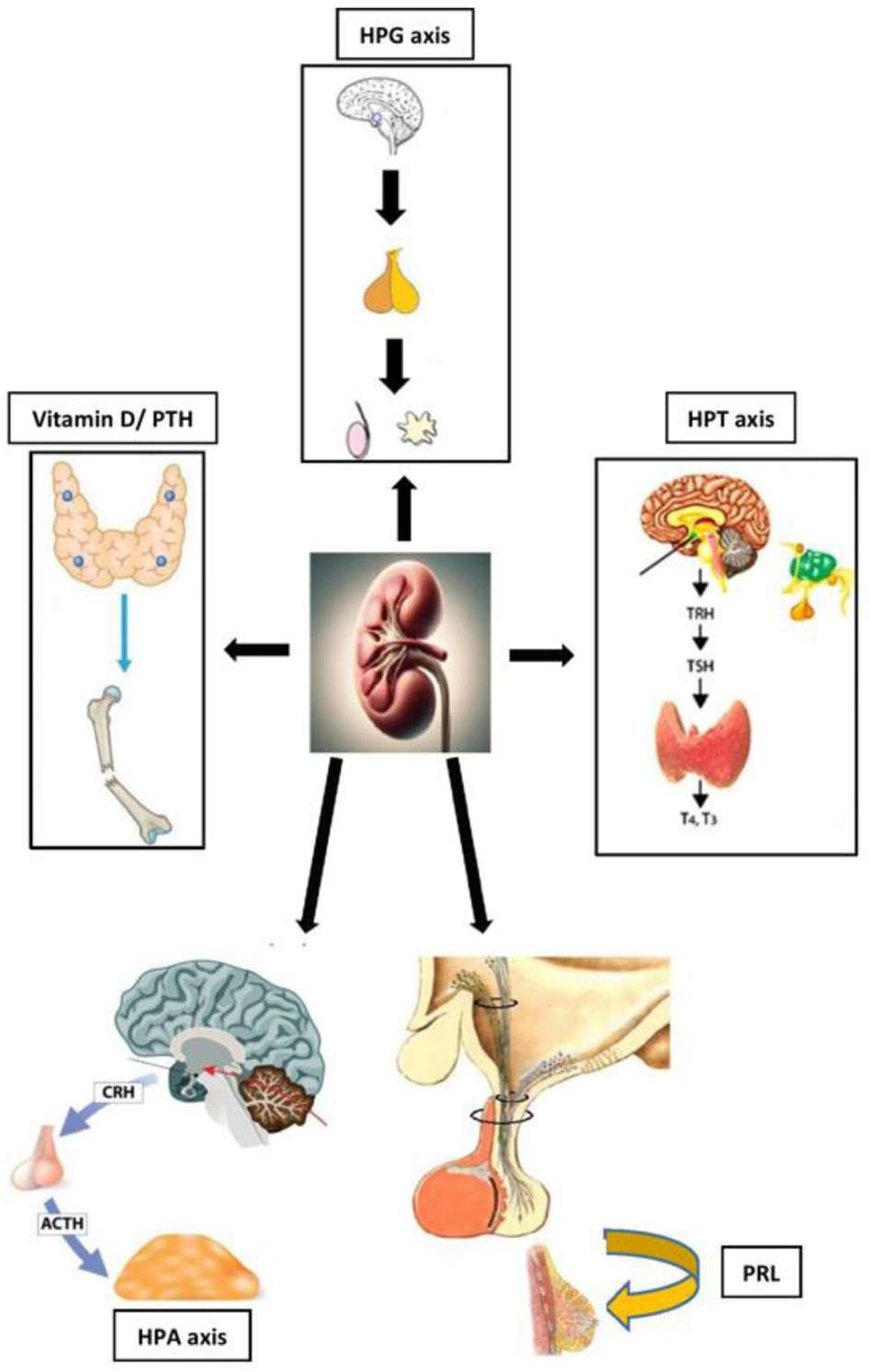
| Testosterone (men) | ↓ | Reduced LH/FSH secretion; chronic uremia; inflammation; ↑ SHBG | Hypogonadism; infertility; loss of muscle mass; decreased libido |
| Estrogens (women) | Cyclic alterations; early menopause | Hypothalamic-pituitary disorders; reduced hormonal clearance | Menstrual irregularities; amenorrhea; reduced fertility |
| Prolactin | ↑ | Reduced renal clearance; hypothalamic dysfunction | Galactorrhea; gynecomastia; sexual dysfunction; increased CV risk |
| Thyroid hormones (FT3, FT4, TSH) | T3 ↓ (low T3 syndrome); TSH normal or ↑ | Altered peripheral deiodination; chronic inflammation | Poor prognosis; ↑ mortality; altered basal metabolism |
| Cortisol | ↑ or dysregulated | Altered clearance and circadian rhythm | Altered glucose metabolism; ↑ CV risk |
| Vitamin D (calcitriol) | ↓ | Reduced renal 1α-hydroxylation | Renal osteodystrophy; ↑ PTH; bone fragility |
| PTH | ↑ | Secondary hyperparathyroidism | Bone and CV alterations |