| World Journal of Nephrology and Urology, ISSN 1927-1239 print, 1927-1247 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, World J Nephrol Urol and Elmer Press Inc |
| Journal website https://wjnu.elmerpub.com |
Original Article
Volume 15, Number 2, April 2026, pages 42-54
Renal Tumors in Young Adults: A Systematic Review and Meta-Analysis of Epidemiology, Clinical Features, Histopathology, and Outcomes Over Two Decades
John Paul R. Tadifaa, b, Rudolfo I. De Guzmana, Kenneth George M. Garciaa
aDepartment of Urology, National Kidney and Transplant Institute, Quezon City, Philippines
bCorresponding Author: John Paul R. Tadifa, Department of Urology, National Kidney and Transplant Institute, Quezon City, Philippines
Manuscript submitted December 4, 2025, accepted February 5, 2026, published online March 28, 2026
Short title: Renal Tumors in Young Adults
doi: https://doi.org/10.14740/wjnu1027
| Abstract | ▴Top |
Background: Renal tumors in young adults are uncommon and less well characterized compared to those in older populations. This systematic review aimed to determine the epidemiology, clinicopathological features, and outcomes of renal tumors in individuals aged 18–45 years.
Methods: Original peer-reviewed cohort studies, randomized controlled trials, and systematic reviews published between 2000 and 2024 were included. Databases searched included PubMed, Scopus, and Web of Science, with the last search conducted in February 2025. Risk of bias was assessed using the Newcastle-Ottawa Scale. A total of 19 studies met the inclusion criteria. Meta-analyses were performed using R (v4.3.1) with a random-effects model.
Results: The pooled proportion of incidental tumor diagnoses was 41% (95% confidence interval (CI): 12–77%), while symptomatic tumors accounted for 42% (95% CI: 23–64%). Clear cell renal cell carcinoma (RCC) was the most prevalent histologic subtype (54%), followed by papillary (12%) and chromophobe (7%). The pooled mean tumor size was 6.84 cm. Recurrence and mortality rates were 4% and 8%, respectively. Most studies reported 5-year cancer-specific survival exceeding 90% for localized RCC. Nephron-sparing surgery was performed in more than half of the patients, supporting its safety and effectiveness in early-stage disease.
Conclusions: Heterogeneity in outcome reporting, small sample sizes in rare subtypes, and possible publication bias were noted. The review supports the importance of early detection, individualized management, and molecular profiling in aggressive subtypes such as Xp11.2 translocation RCC.
Keywords: Renal tumors; Young adults; Renal cell carcinoma; Epidemiology; Clinicopathologic features; Nephron-sparing surgery; Systematic review; Xp11.2 translocation RCC
| Introduction | ▴Top |
Late adolescents and young adults represent a special subset of the population when it comes to urological malignancies such as renal tumors [1]. Renal cell carcinoma (RCC) is predominantly diagnosed among older adults, with a median age of 64 years, and individuals younger than 40 years account for up to 7% of renal tumors [2]. Renal tumors in young adults, however, often present with distinct clinicopathological features, including a higher prevalence of hereditary syndromes and generally better prognosis compared with older adults [1, 2].
Recent studies highlight that early detection and tailored management strategies are essential in this population [1, 2]. The incidence of RCC among young patients has been increasing, likely driven by the widespread use of imaging modalities such as ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI) [2]. Despite this trend, the clinical profile of renal tumors in young adults remains underexplored, with many cases diagnosed incidentally during routine health evaluations or workups for unrelated conditions [1]. Unlike other urologic diseases, there are currently no established guidelines for screening this population, partly due to cost considerations, limited causal evidence, and insufficient understanding of associated risk factors [1, 2].
This systematic review aims to provide a comprehensive overview of the epidemiology, clinicopathologic features, and clinical outcomes of renal tumors in young adults. By synthesizing two decades of available literature, this review seeks to identify patterns that may guide improved diagnostic and therapeutic approaches for this unique patient population. The general objective of the study was to determine the overall behavior of renal tumors among young adults. The specific objectives were to identify the epidemiology, clinicopathologic features, and clinical outcomes/prognosis.
| Materials and Methods | ▴Top |
The study was a systematic review. Ethical approval was not required because all analyses were based on published studies and did not involve new data collection from human participants. Studies eligible for inclusion must meet the following criteria: 1) original research articles (cohort studies (retrospective/prospective), randomized controlled trials (RCTs), and systematic reviews) published in peer-reviewed journals within 24 years (2000–2024); 2) studies investigating epidemiology, clinicopathologic features, clinical outcome, and prognosis of renal tumors/masses in young adults (18–45 years old); and 3) availability of sufficient data for systematic review/meta-analysis. Studies on patients < 18 or > 45 years old, case reports, conference abstracts, and those without relevance to the study outcomes were excluded.
Information sources
The following electronic databases were searched for all available years: PubMed, Scopus, Web of Science.
Search strategy
A comprehensive literature search was conducted in PubMed, Scopus, and Web of Science for studies published between 2000 and 2024. Search terms combined controlled vocabulary and free-text keywords related to renal tumors (e.g., renal mass, renal tumor, renal cancer, renal carcinoma) and young adults (e.g., young adult, early onset). The final search was performed in February 2025. Full database-specific search strategies and results are provided in Supplementary Material 1 (wjnu.elmerpub.com).
Selection process
The principal investigator reviewed all potential studies for inclusion against the eligibility criteria. The investigator examined the title, abstract, and, where necessary, the full text of studies to assess if they are eligible for inclusion.
Data collection process
Three reviewers used a standard form (101HR_Form 5) to independently extract study characteristics and outcome data from the studies. Discrepancies were checked against the original article. Disagreements in values were settled through adjudication of authors.
During the initial screening phase, automation tools were used to facilitate record management and exclusion. Database-embedded duplicate detection functions within PubMed, Scopus, and Web of Science, as well as citation-manager–based duplicate removal, were applied to exclude duplicate records. Records were automatically excluded for the following reasons: duplicate citations, non-renal malignancies, non-human studies, non-English language publications, pediatric-only populations, and publication types such as conference abstracts, editorials, or commentaries. The remaining 595 records underwent manual title and abstract screening by two independent reviewers. Studies were excluded at this stage if they did not meet predefined age-related inclusion criteria, lacked extractable subgroup data for young adults, or did not report relevant clinical or outcome data.
Data items
All reported outcome measures were extracted from the studies. Primary outcomes included cancer-specific survival (CSS), overall survival (OS), and tumor recurrence. Secondary outcomes, such as renal function preservation, treatment modality effectiveness, and additional clinical and pathological parameters, were also recorded. When multiple measures (e.g., various time points) were provided by a study, the most comprehensive and primary result was prioritized.
In addition to outcomes, data on study characteristics were extracted, including setting, design, inclusion and exclusion criteria, and participant demographics (e.g., age and sex). Additional clinical parameters, such as tumor size, histopathologic findings, and treatment details, were recorded to facilitate subgroup analyses and contextual interpretation. For variables with missing or ambiguous information, assumptions were made based on the study’s inclusion criteria or were clarified through further review of the study methods.
Study risk of bias assessment
The risk of bias in the included studies was assessed using the Newcastle-Ottawa Scale (NOS) (Table 1), a validated tool for evaluating the quality of non-randomized studies in meta-analyses [1, 3–17]. The NOS assesses studies across three domains: selection, comparability, and outcome. Based on the total NOS score, studies were categorized as having low (7–9 stars), moderate (4–6 stars), or serious (0–3 stars) risk of bias.
 Click to view | Table 1. Risk of Bias Assessment of Included Studies Using the Newcastle–Ottawa Scale (NOS) |
Three reviewers independently evaluated each study, and any disagreements in scoring were resolved through discussion and consensus. No automation tools were used in the assessment process. No adaptations were made to the NOS, and no new tools were developed for this review.
Effect measures
For the synthesis and presentation of outcomes across the included studies, various effect measures were employed, depending on the type of outcome.
For binary outcomes (e.g., survival status, presence of metastasis, recurrence), the effect measures included: 1) CSS and OS rates, often expressed as percentages at 1-, 5-, and 10-year intervals; and 2) hazard ratios (HRs) with 95% confidence intervals (CIs) used in studies [1, 3–6] to identify prognostic factors for survival outcomes.
For time-to-event outcomes, studies employed: progression-free survival (PFS) and recurrence-free survival (RFS), expressed as Kaplan–Meier survival curves, survival percentages at defined time points, or HRs [1, 6].
For continuous outcomes (e.g., tumor size, serum creatinine levels), the studies presented: mean ± standard deviation (SD) or median with interquartile range (IQR) values. These were primarily used for tumor dimension comparisons, renal function before and after surgery, and duration of follow-up [7, 8].
No re-expression of effect measures (e.g., converting HRs to absolute risk reductions) was applied in this review. Similarly, no thresholds for clinically meaningful effects (e.g., minimally important differences) were explicitly defined by the included studies or by this review. Thus, interpretation of effect sizes was contextual and based on clinical relevance as reported by individual studies.
Justification for these effect measures aligns with standard practice in clinical prognosis and surgical outcome studies which allows for consistent comparisons across observational cohorts.
Synthesis method
Eligible studies were confirmed through title/abstract and full-text screening against predefined criteria and then summarized in Supplementary Materials 2 and 3 (wjnu.elmerpub.com) capturing study design, participant characteristics, treatment approach, and outcomes of interest.
For outcomes with sufficient and comparable data, we performed random-effects meta-analyses in R version 4.3.1: the inverse-variance (IV) method for continuous outcomes and a generalized linear mixed model (GLMM) for pooled proportions.
A qualitative synthesis was performed for all included studies. For outcomes with sufficient data, meta-analyses were conducted using R version 4.3.1. Two separate random-effects models were employed: 1) for continuous outcomes, the IV method with a random-effects model was used; 2) for binary outcomes, a GLMM approach was applied.
The restricted maximum likelihood (REML) estimator was used to calculate between-study variance (τ2), and Hartung-Knapp CIs were applied for pooled estimates.
Statistical heterogeneity was evaluated using the I2 statistic, tau-squared (τ2), and the Chi-squared (Q) test. Visual inspection of forest plots further supported the assessment of heterogeneity across studies.
Reporting biases assessment
To assess the risk of bias due to missing results in the synthesis (i.e., reporting bias), funnel plots were generated using R version 4.3.1 for outcomes that included 10 or more studies, specifically for clear cell histology, papillary histology, chromophobe histology, and overall mortality.
The funnel plots compared the standard error against the logit-transformed proportions. Asymmetry in the funnel plots was visually evaluated to detect potential small-study effects or publication bias.
Certainty assessment
We assessed the certainty of evidence using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework, following the GRADE Handbook guidance [18]. Since all included primary studies were non-randomized observational cohorts, certainty for every quantitative outcome began at low certainty.
Two reviewers, working independently, judged the five GRADE domains: risk-of-bias, inconsistency, indirectness, imprecision, and publication bias for each outcome. Disagreements were resolved by discussion or, if needed, adjudication by a third reviewer. Risk-of-bias was derived from the NOS scores of the contributing studies and downgraded one level when ≥ 50% of participants came from studies at high or unclear risk.
Inconsistency was evaluated with the I2 statistic and inspection of forest plots; I2 > 50% without an obvious clinical explanation triggered a one-level downgrade, while I2 > 75% prompted a two-level downgrade. Indirectness considered whether the population (18–45 years), exposure (sporadic or hereditary renal tumors), and outcome definitions matched our review question; no downgrades were necessary. Imprecision was judged against a minimally important difference of 10 percentage points for risk outcomes and 2 cm for mean tumor size. We downgraded when 95% CIs crossed this threshold or when total events were < 300. We prespecified that observational evidence could be upgraded for a large effect (relative risk (RR) ≤ 0.5 or ≥ 2.0) observed across multiple studies or for an exposure–response gradient; neither criterion was met, so no upgrades were applied.
For each critical outcome (incidence trend, histologic distribution, tumor size, presentation mode, recurrence, OS, and CSS), we produced a GRADE evidence profile and a “summary of findings” table. Certainty was categorized as high, moderate, low, or very low and interpreted according to GRADE guidance.
Reporting guidelines
This systematic review was conducted and reported in accordance with the PRISMA 2020 statement [19].
| Results | ▴Top |
The systematic search across three databases, PubMed (n = 618), Scopus (n = 991), and Web of Science (n = 3,867), yielded a total of 5,476 records. After removing 671 duplicates, 4,138 records were excluded by automation tools, and 595 were removed for other reasons, leaving 72 records for screening.
Out of these, four full-text reports were sought for retrieval, and all were successfully retrieved. Following full-text assessment of 22 reports, three were excluded due to lack of subgroup analysis specific to individuals aged 18–45 years.
Ultimately, 19 studies were included in the review. The complete selection process is summarized in Figure 1. No ongoing studies or associated reports were identified during the review process.
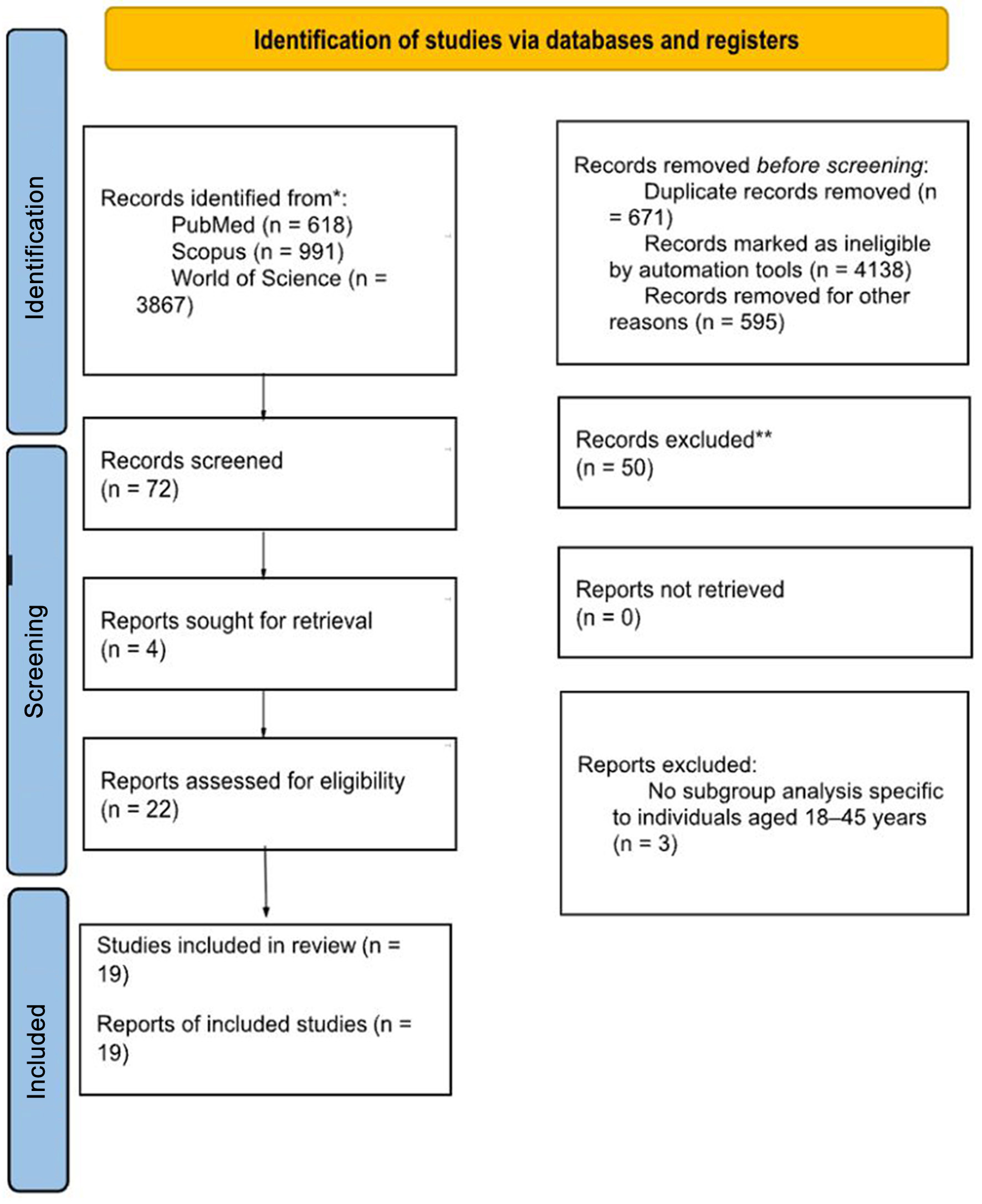 Click for large image | Figure 1. PRISMA 2020 flow diagram of study selection. Flow diagram showing identification, screening, eligibility assessment, and inclusion of studies evaluating renal tumors in young adults (18–45 years). |
Study characteristics are shown in Supplementary Materials 2 and 3 (wjnu.elmerpub.com).
Risk of bias in studies
The included studies were assessed for risk of bias using the NOS, which evaluates three domains: selection, comparability, and outcome. The total NOS score determines whether a study is categorized as having a low (7–9 stars), moderate (4–6 stars), or serious (0–3 stars) risk of bias. NOS scores of individual primary studies are presented in Table 1.
Selection bias
The selection domain evaluates the representativeness of the exposed cohort, selection of the non-exposed cohort, ascertainment of exposure, and demonstration that the outcome was not present at the start of the study.
Representativeness of the exposed cohort
Most studies [1, 3, 8] had somewhat representative exposed cohorts, while others [9, 10] were either selective (e.g., targeting specific professions like nurses) or provided no description.
A lack of representativeness can introduce selection bias, limiting the generalizability of findings.
Selection of the non-exposed cohort
Several studies [7, 11, 12] did not provide details about their control groups, while others [3] ensured that controls were drawn from the same population.
A failure to select an appropriate control group increases the likelihood of selection bias.
Ascertainment of exposure
Many studies [1, 13, 14] relied on secure records (e.g., medical or surgical records), reducing bias.
Others [10, 12] used self-reports, which introduce recall bias and reduce reliability.
Demonstration that the outcome was not present at the start of the study
Nearly all studies met this criterion, ensuring that outcomes developed during the study period rather than pre-existing.
Comparability bias
Comparability assesses whether studies controlled for confounding variables.
Control for confounders
While some studies [1, 3] controlled for at least one confounding variable, others [9, 12] did not account for any, increasing the risk of confounding bias.
Few studies adjusted for additional confounders, which may lead to residual confounding effects.
Outcome bias
The outcome domain examines assessment methods, follow-up duration, and completeness of follow-up.
Assessment of outcome
Many studies [1, 6, 15] used independent blind assessment or record linkage, reducing bias.
Some studies [10, 11] relied on self-reports, which introduce information bias.
Follow-up duration
Most studies had adequate follow-up durations, ensuring that outcomes had sufficient time to develop.
Loss to follow-up
Some studies [5, 12] had incomplete follow-up data or lacked clear reporting on lost participants, increasing attrition bias.
Overall quality assessment
Overall quality assessment was conducted based on the NOS scores. Four studies [1, 3, 6, 8] were rated as having low risk of bias (7–9 stars), three studies [9, 11, 12] as having moderate risk of bias (4–6 stars), and none of the included studies were categorized as having serious risk of bias (0–3 stars).
The majority of included studies were rated low to moderate risk of bias, with strong methodological rigor in exposure assessment and outcome evaluation. However, selection bias and inadequate confounder control were the most common sources of bias, particularly in studies that lacked well-defined control groups or used self-reported data.
To provide additional context, we discussed in greater detail the studies with the largest sample sizes and most complete outcome reporting, while all included studies contributed proportionally to the pooled estimates.
Results of individual studies are shown in Supplementary Materials 2 and 3 (wjnu.elmerpub.com).
Results of syntheses
The pooled proportion of incidental tumor diagnoses was 41% (95% CI: 12–77%) (Fig. 2). The high heterogeneity (I2 = 95%) may reflect differences in imaging utilization, healthcare access, and referral thresholds across settings.
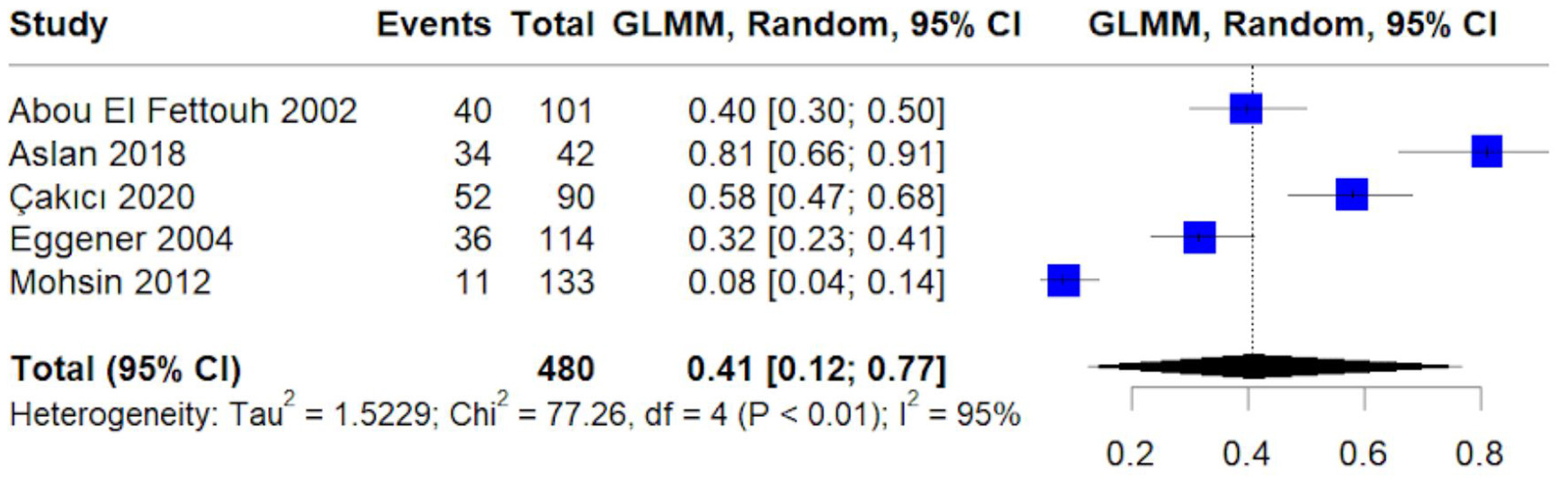 Click for large image | Figure 2. Forest plot of pooled proportion of incidental tumor diagnosis. Random-effects generalized linear mixed model (GLMM) estimating the pooled proportion of tumors detected incidentally. Proportions are presented with 95% confidence intervals. |
The pooled proportion of symptomatic tumor diagnoses was 42% (95% CI: 23–64%) (Fig. 3). These cases were typically detected due to hematuria, flank pain, or other symptoms, and the heterogeneity (I2 = 89%) likely reflects variation in symptom definitions and diagnostic pathways.
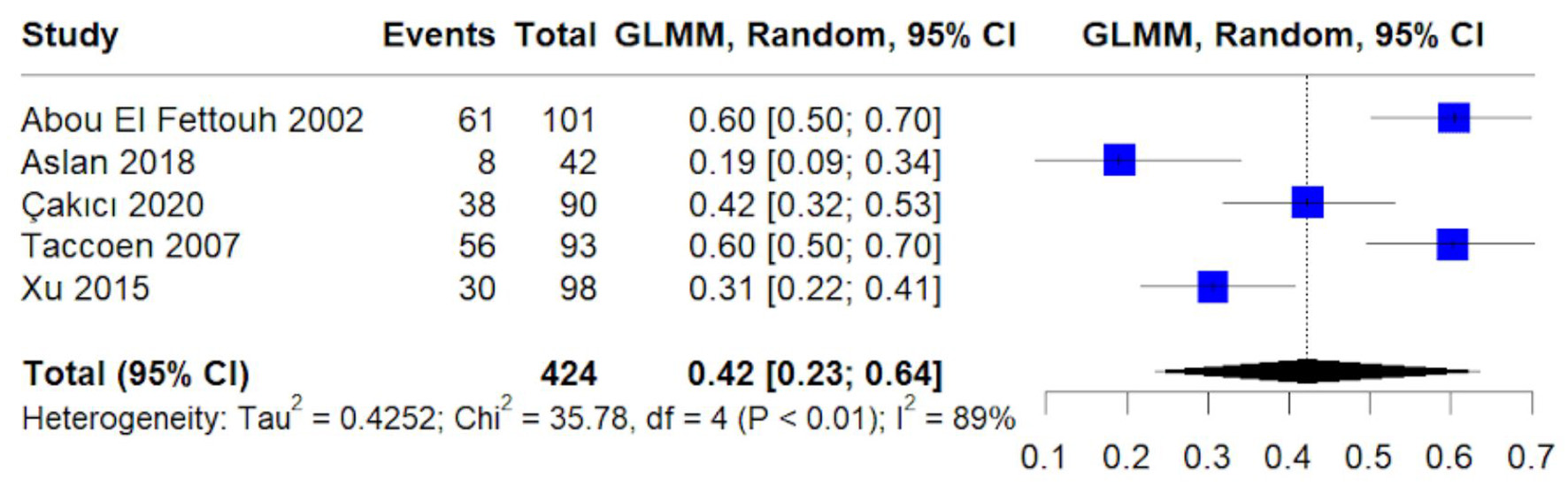 Click for large image | Figure 3. Forest plot of pooled proportion of symptomatic tumor diagnosis. Random-effects GLMM estimating pooled symptomatic presentation rates with 95% confidence intervals. |
The pooled mean tumor size was 6.84 cm (95% CI: 3.21–10.47 cm) (Fig. 4). Substantial heterogeneity (I2 = 90%) suggests variability in case mix and timing of detection across cohorts.
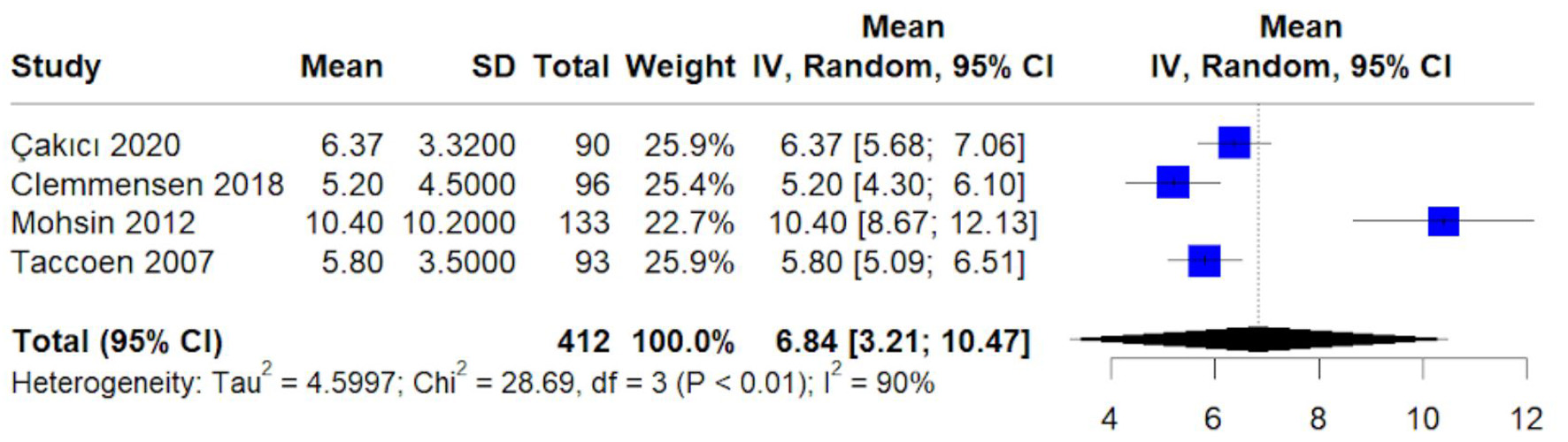 Click for large image | Figure 4. Forest plot of pooled mean tumor size (cm). Random-effects inverse-variance model showing pooled mean tumor size with 95% confidence intervals. Heterogeneity assessed using I2 and τ2 statistics. |
Clear cell RCC was the most prevalent subtype, accounting for 54% of cases (95% CI: 40–67%) (Fig. 5). Heterogeneity was high (I2 = 97%), consistent with differences in population characteristics and histologic reporting across studies.
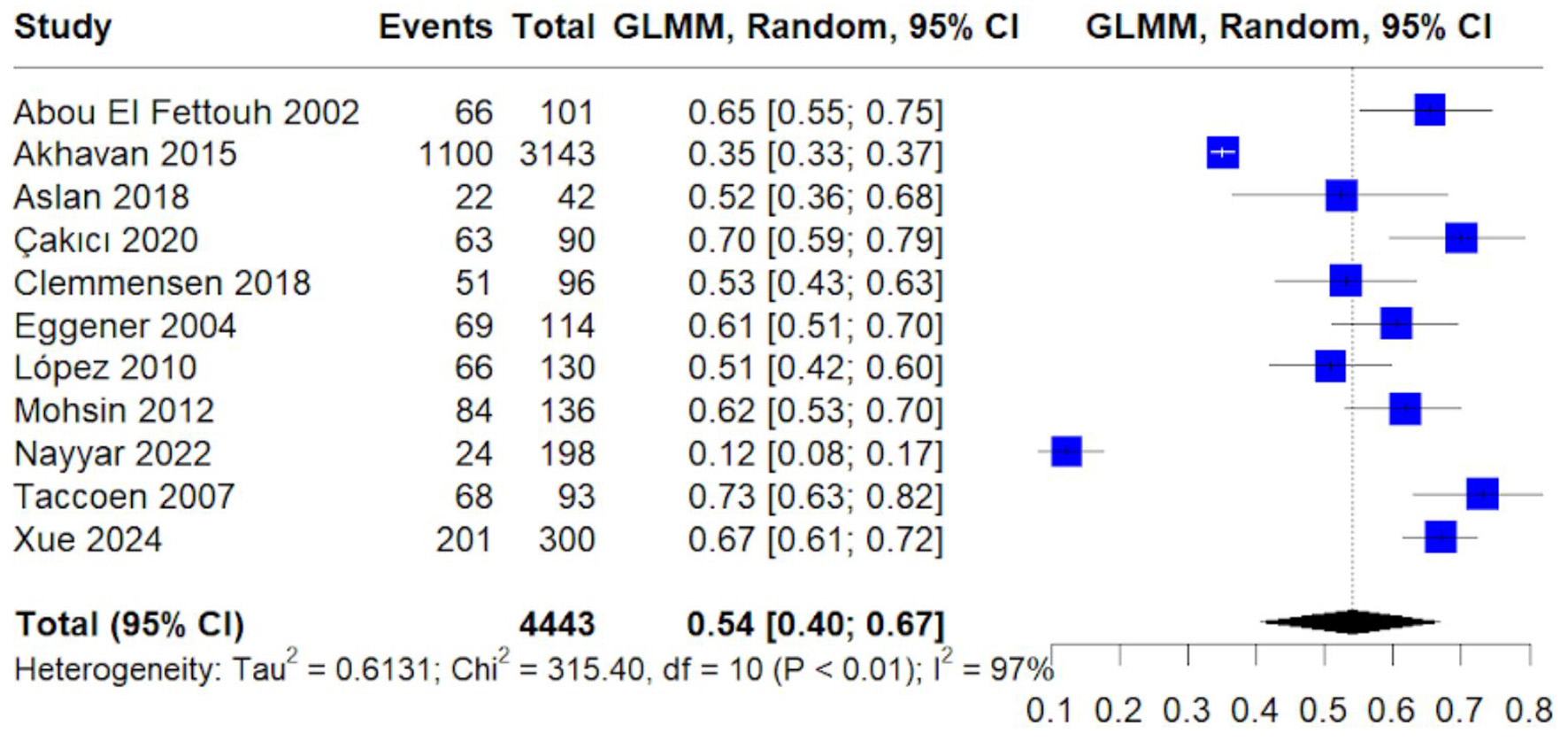 Click for large image | Figure 5. Forest plot of pooled proportion of clear cell histology. Random-effects GLMM estimating prevalence of clear cell renal cell carcinoma among young adults. |
Papillary histology accounted for 12% of cases (95% CI: 9–16%) (Fig. 6). Heterogeneity was moderate to high (I2 = 83%), likely reflecting differences in cohort composition and histologic classification across studies.
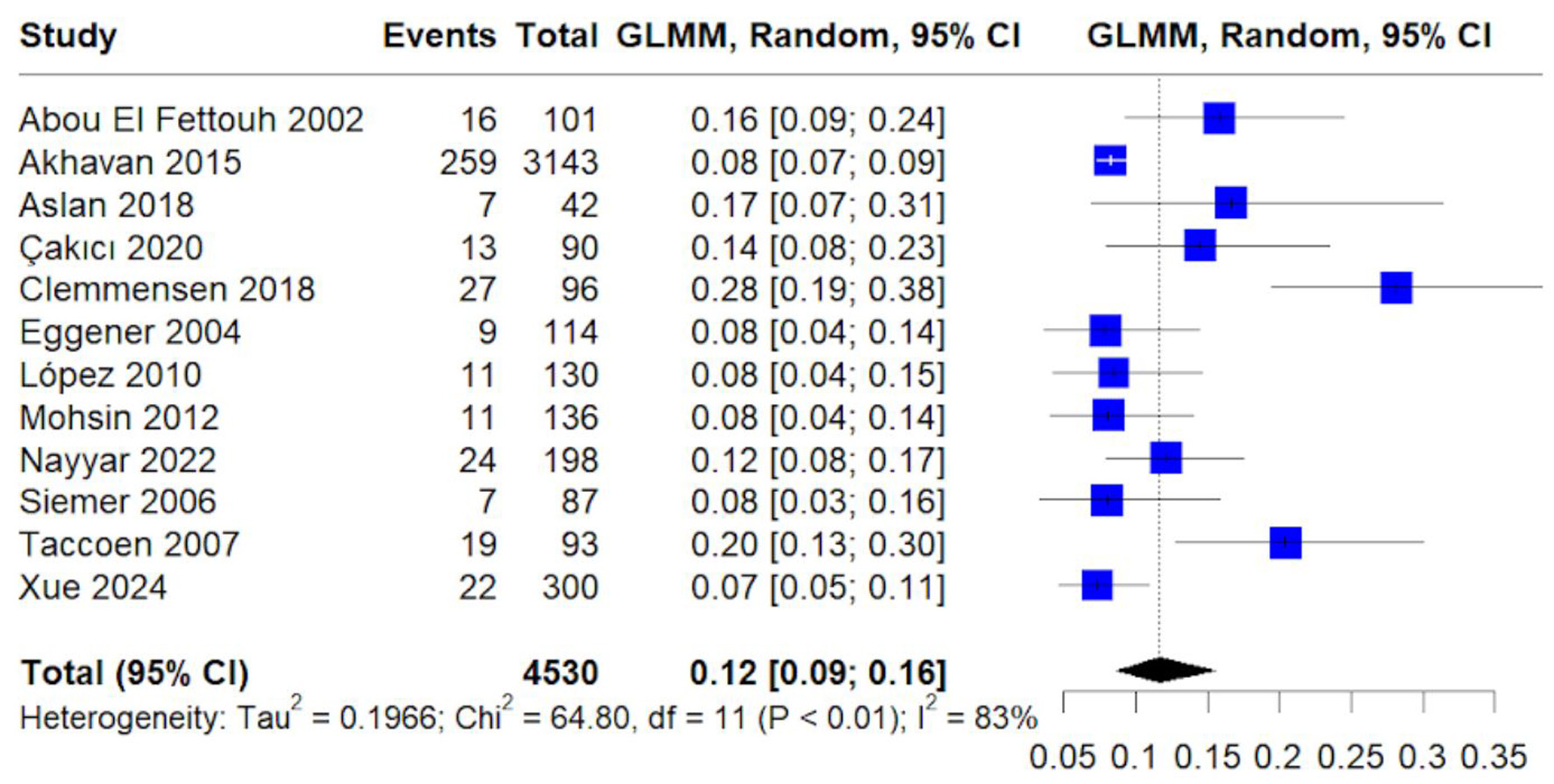 Click for large image | Figure 6. Forest plot of pooled proportion of papillary histology. Random-effects GLMM estimating prevalence of papillary renal cell carcinoma. |
Chromophobe histology accounted for 7% of cases (95% CI: 5–10%) (Fig. 7). Heterogeneity was moderate (I2 = 62%), suggesting less between-study variability than other pooled histology outcomes.
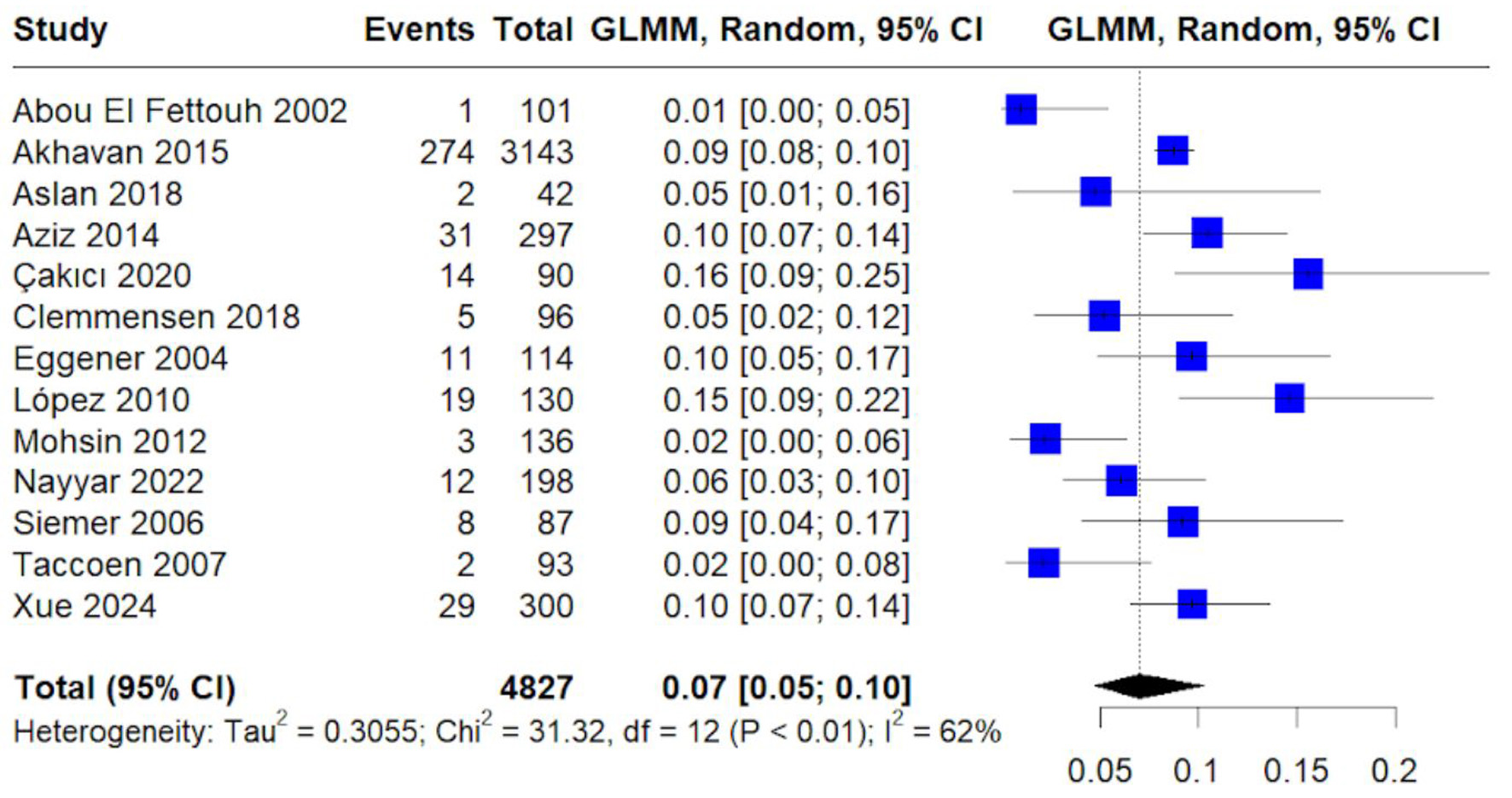 Click for large image | Figure 7. Forest plot of pooled proportion of chromophobe histology. Random-effects GLMM estimating prevalence of chromophobe renal cell carcinoma. |
The pooled recurrence rate was 4% (95% CI: 1–20%) (Fig. 8). Heterogeneity was high (I2 = 83%), likely related to differences in follow-up duration and recurrence definitions across cohorts.
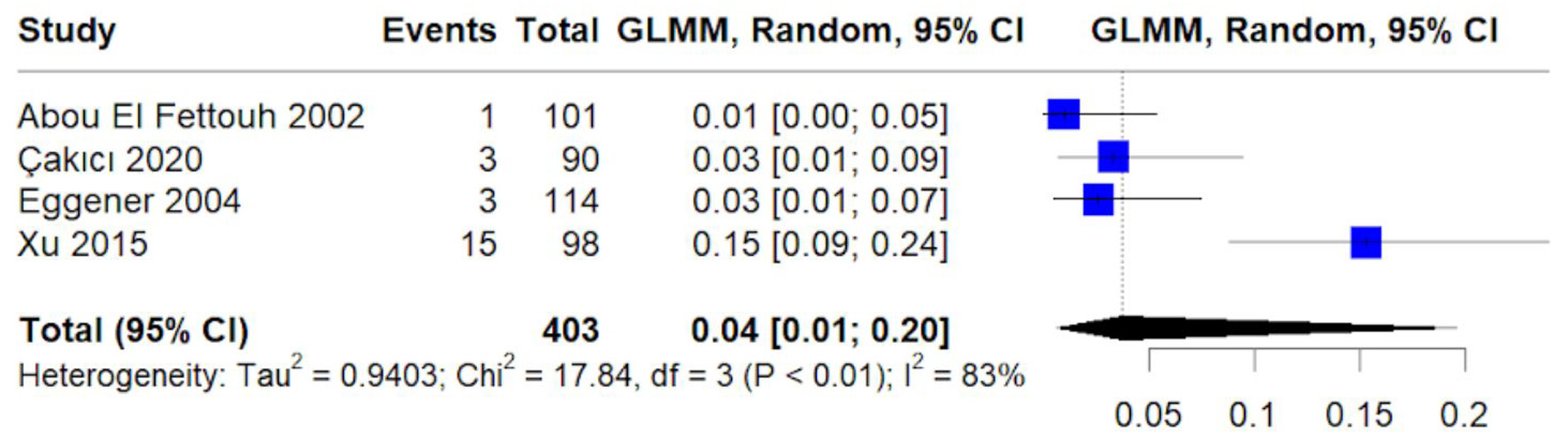 Click for large image | Figure 8. Forest plot of pooled proportion of tumor recurrence. Random-effects GLMM estimating pooled recurrence rate following treatment. |
The pooled overall mortality was 8% (95% CI: 4–15%) (Fig. 9). High heterogeneity (I2 = 87%) suggests variability in baseline risk, stage distribution, and duration of follow-up.
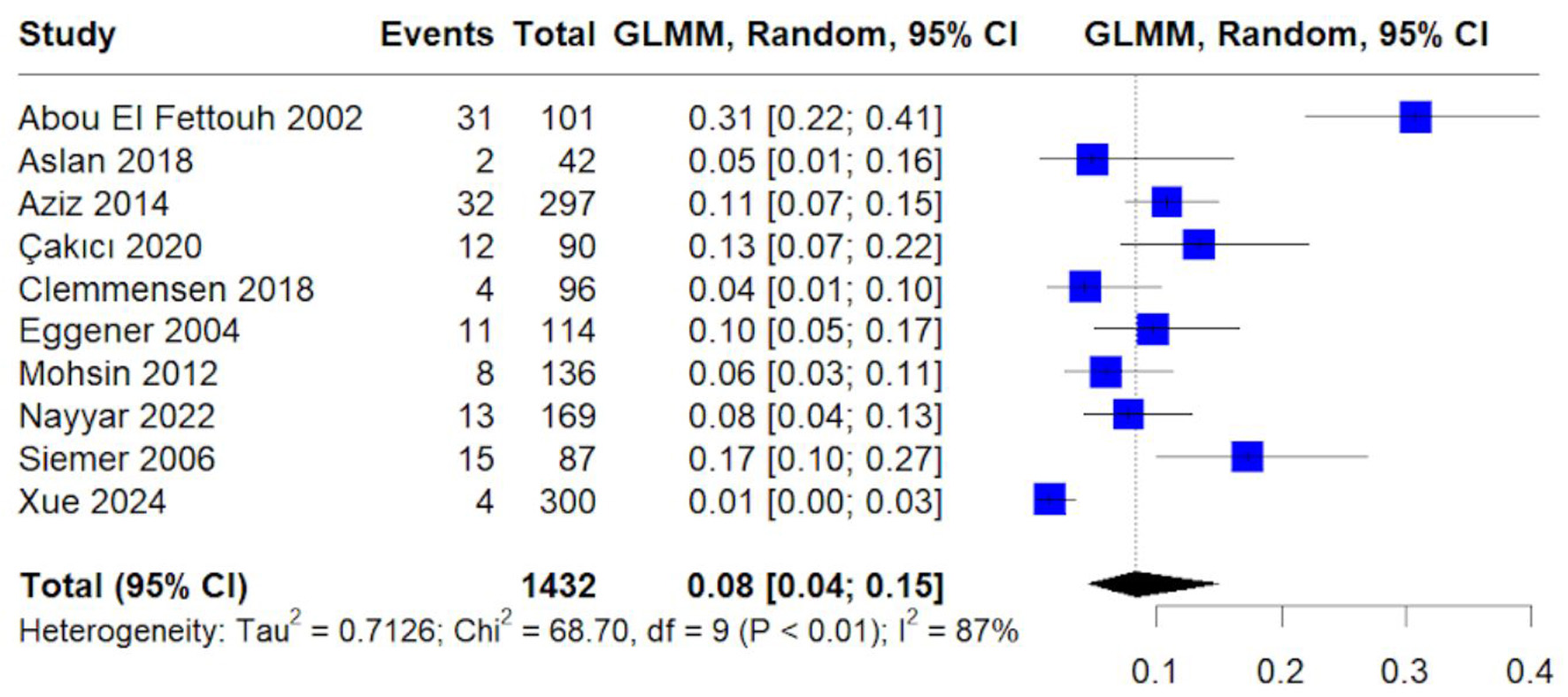 Click for large image | Figure 9. Forest plot of pooled proportion of overall mortality. Random-effects GLMM estimating pooled mortality rate among included cohorts. |
Reporting biases (due to missing results)
To assess the risk of bias due to missing results (i.e., reporting biases), funnel plots were generated for syntheses with at least 10 included studies. These plots were used to visually evaluate small-study effects, one potential indicator of publication or selective reporting bias.
Funnel plots were generated for the following outcomes: clear cell histology (Fig. 10), papillary histology (Fig. 11), chromophobe histology (Fig. 12), and overall mortality (Fig. 13).
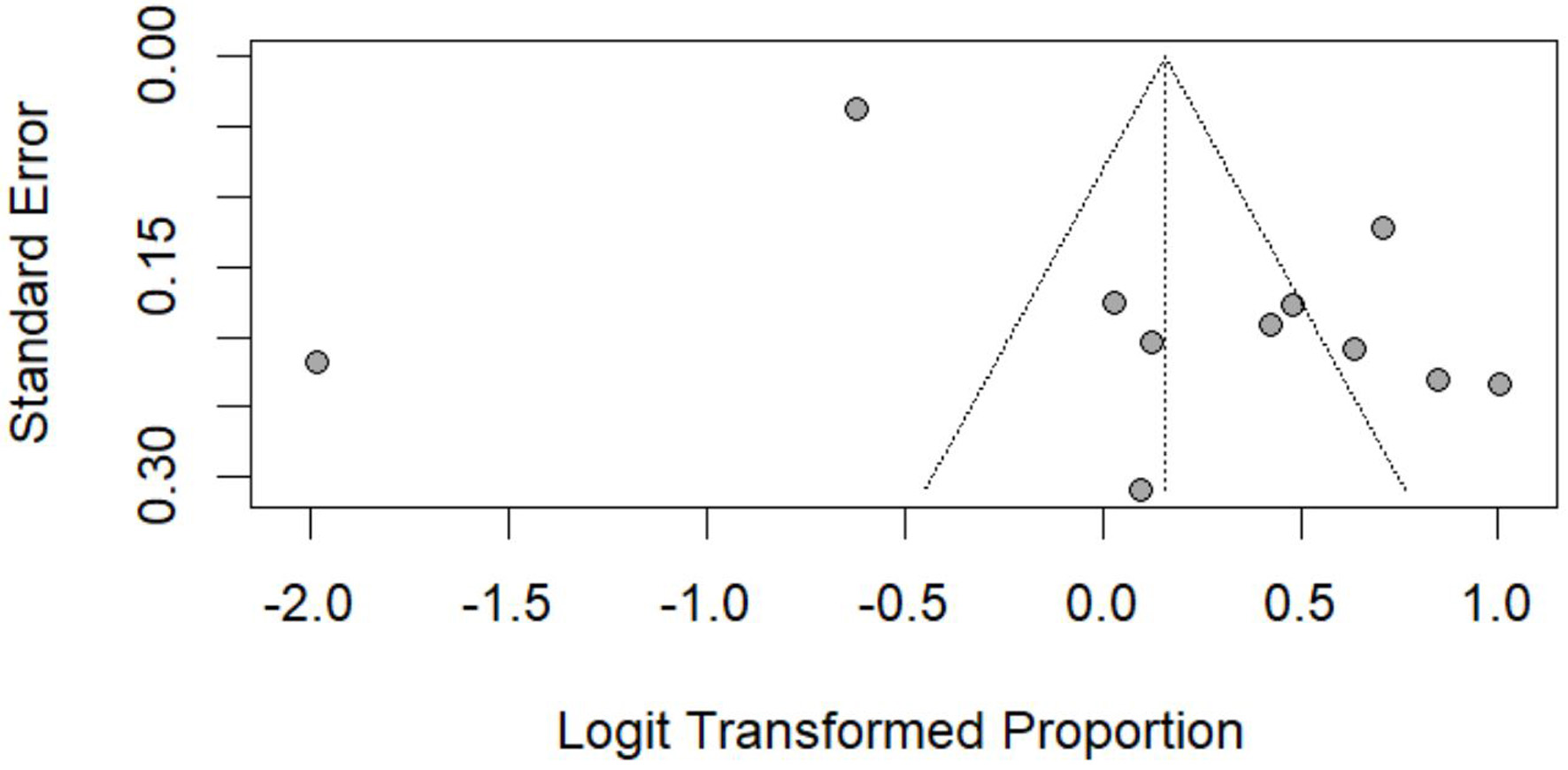 Click for large image | Figure 10. Funnel plot assessing publication bias for clear cell histology. Funnel plot of standard error versus logit-transformed proportion. Symmetry assessed visually. |
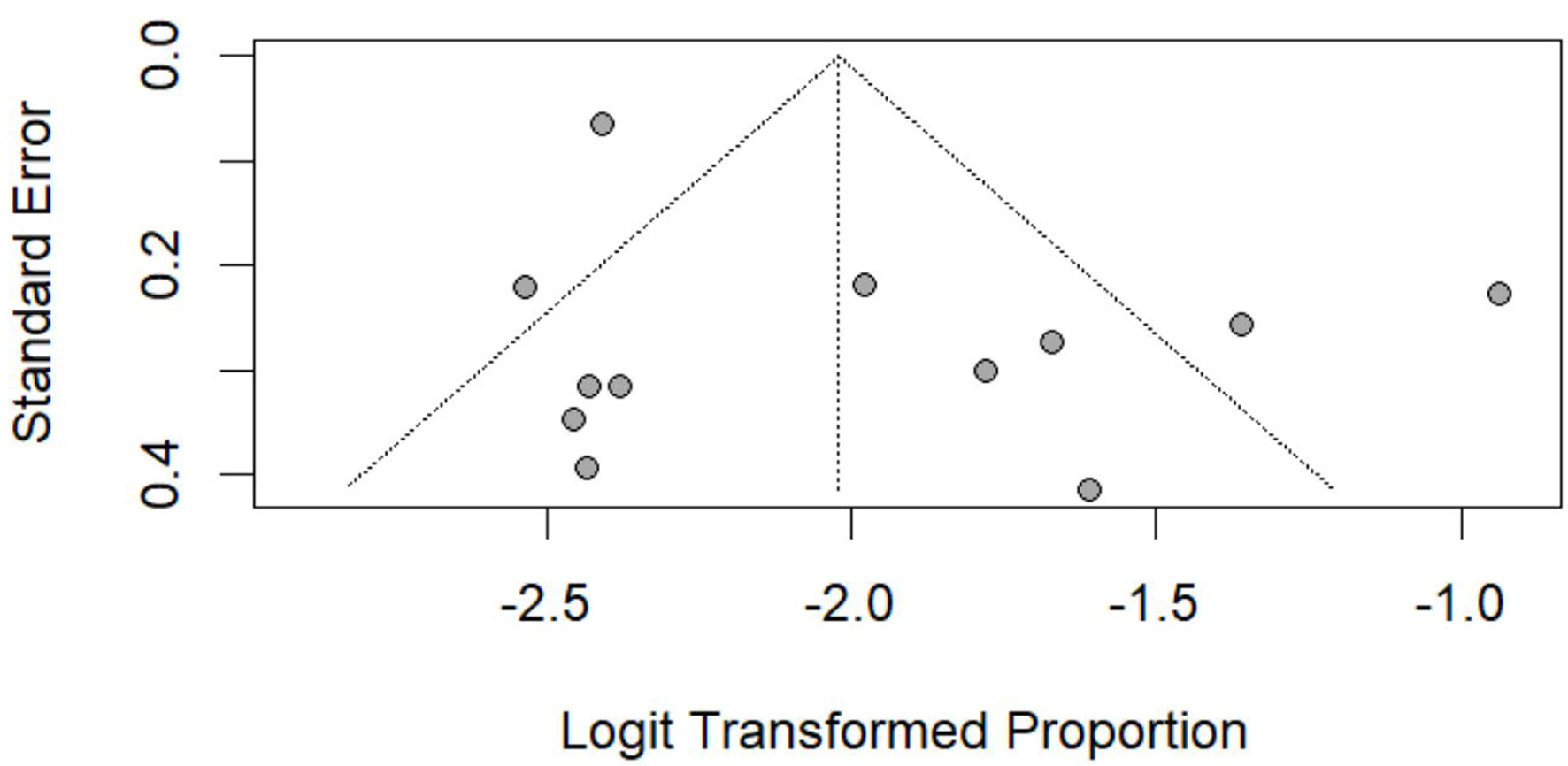 Click for large image | Figure 11. Funnel plot assessing publication bias for papillary histology. Standard error plotted against logit-transformed pooled proportions. |
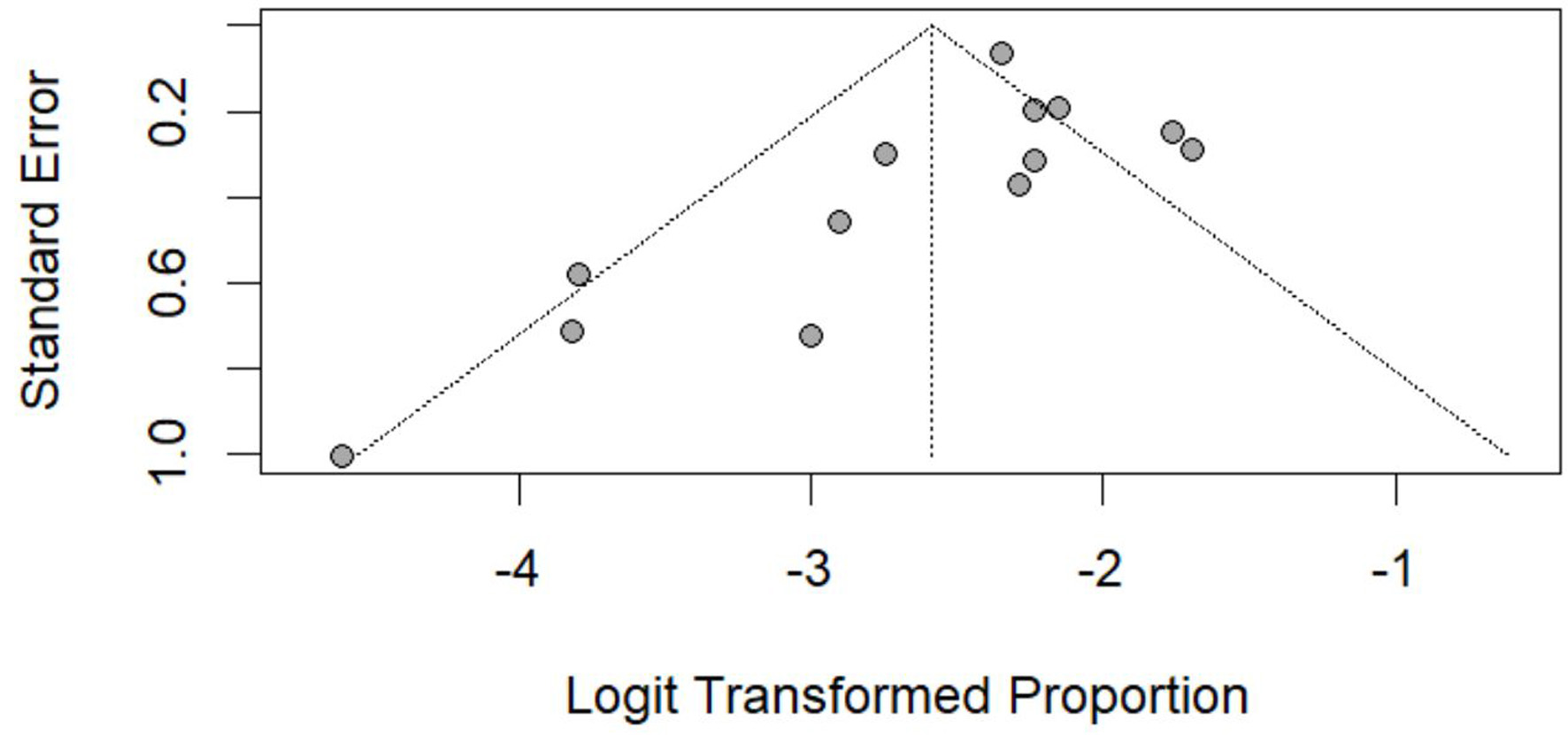 Click for large image | Figure 12. Funnel plot assessing publication bias for chromophobe histology. Visual assessment of small-study effects. |
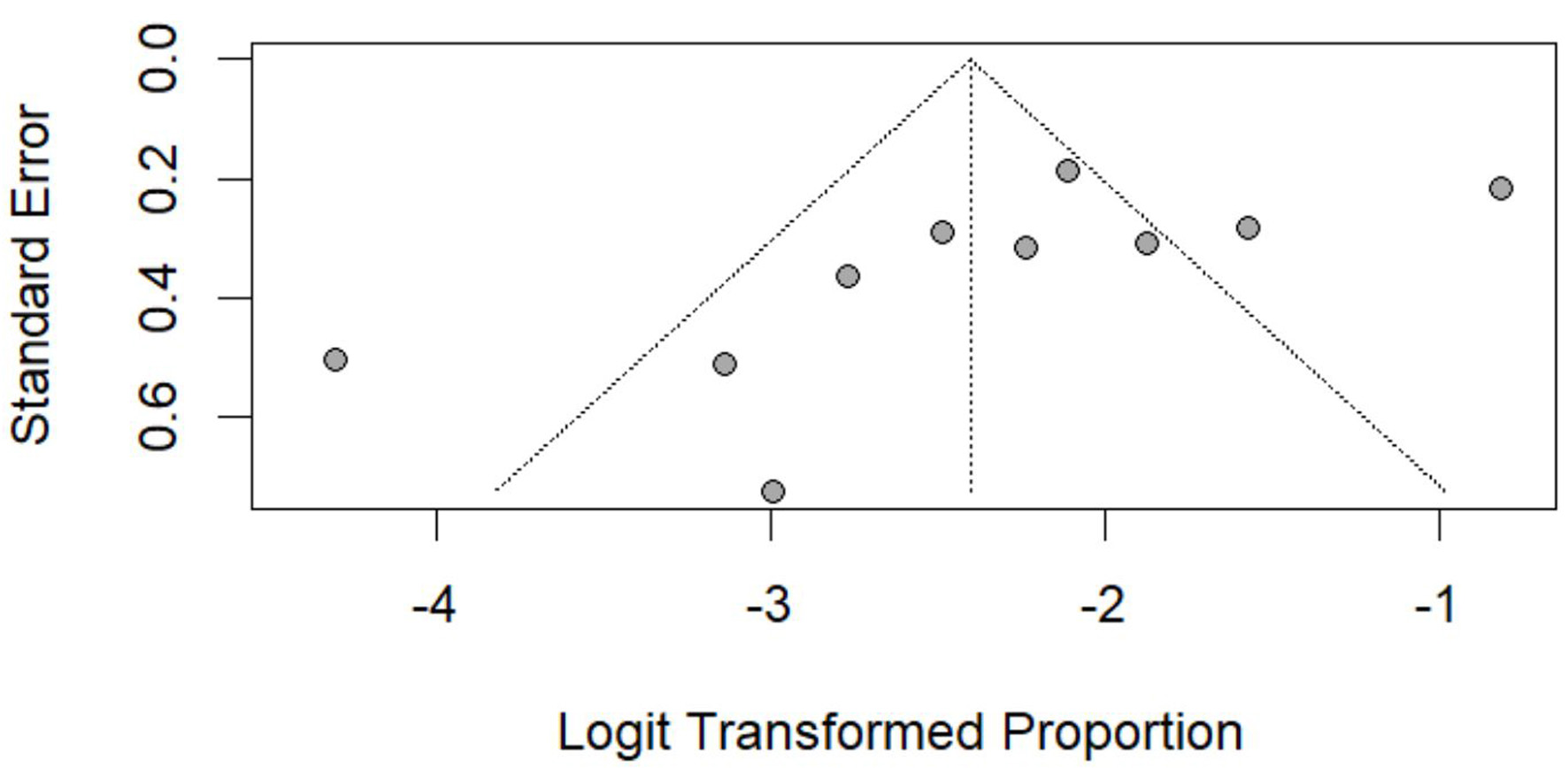 Click for large image | Figure 13. Funnel plot assessing publication bias for overall mortality. Standard error plotted against logit-transformed pooled proportions. |
The plots compared standard error (as a measure of precision) against the logit-transformed proportions (as effect estimates). In each funnel plot, the y-axis represents the standard error (precision) and the x-axis represents the logit-transformed pooled proportion for the specified outcome (e.g., clear cell, papillary, chromophobe histology, or overall mortality).
Funnel plot inspection for clear cell histology suggested some asymmetry, which may reflect small-study effects and/or reporting bias (Fig. 10).
The funnel plot for papillary histology showed mild asymmetry among smaller studies (Fig. 11).
The funnel plot for chromophobe histology appeared largely symmetrical, suggesting minimal evidence of publication bias (Fig. 12).
The funnel plot for overall mortality was generally symmetrical, with slight dispersion among smaller studies (Fig. 13).
Certainty of evidence
Using the GRADE approach, we judged each outcome separately and then summarized the review’s certainty by the lowest-rated critical outcome. All evidence derives from retrospective cohort studies, so every outcome began at LOW certainty and was routinely downgraded for serious risk-of-bias (incomplete adjustment for confounding) and very serious inconsistency (I2 often > 75%); several outcomes were further downgraded for imprecision caused by wide confidence intervals.
As a result, incidence trends and histology mix were rated low certainty, whereas the key clinical endpoints: CSS, recurrence, tumor size, mode of presentation, and stage/grade distribution fell to very low certainty. Accordingly, the overall strength of the evidence supporting conclusions about the behavior of renal tumors in young adults is very low, meaning that our confidence in the effect estimates is limited and the true effects are likely to differ substantially.
| Discussion | ▴Top |
Key findings
Previous narrative and systematic reviews have described the epidemiology, clinicopathologic features, and management considerations of RCC in pediatric and young adult populations, including the role of nephron-sparing surgery and long-term surveillance strategies [2, 20, 21].
Our review consolidates evidence from 17 single-center cohorts and two US population datasets to characterize renal tumors in young adults. Across all studies, a pronounced male excess is evident (male/female ≈ 1.7:1 [7] and 1.6:1 [13]), mirroring the sex ratio reported in national cancer statistics. Roughly 41% of cancers were discovered incidentally, usually on ultrasound or CT performed for unrelated complaints, while 42% presented with hematuria, flank pain or a palpable mass. The extremes of individual series (8% incidental [11] vs. 81% [9]) illustrate wide practice-pattern heterogeneity. Incidentally detected tumors were smaller and predominantly stage T1, whereas symptom-driven cases were often larger, higher stage and linked to poorer survival, as reported by Mohsin et al [11] and Siemer et al [4].
Clear-cell RCC dominated, accounting for about 54% of lesions (e.g., 55% [16] and 51% [12]). Papillary (about 12%) and chromophobe (about 7%) subtypes together comprised about one-third of cases. Several cohorts highlighted aggressive but uncommon entities: Xp11.2 translocation RCC occurred in up to 19% of tumors in the Chinese series of Xu et al [14] and carried a four-fold higher cancer-specific mortality. Likewise, sarcomatoid features present in 4–6% of young adult tumors were consistently linked to early recurrence and death [4, 8].
Survival in this age group is generally favorable. Five-year CSS exceeded 90% in Taccoen et al [5] (young cohort) and 92% in Aziz et al [13], while large US registry data showed a 5-year CSS of 89.9%. Nevertheless, pooled recurrence was about 4%, and stage IV or nodal disease, present in 5–9% of cases [1, 6], portended markedly worse outcomes. Classic prognosticators (stage ≥ T3, node-positive status, Fuhrman/ISUP grade 3-4) retained their predictive power even after age adjustment in multivariable models from Siemer et al [4] and Xu et al [14].
Surgery remains the mainstay of cure. The apparent survival advantage associated with partial nephrectomy likely reflects selection bias, as patients selected for nephron-sparing surgery more often have smaller, organ-confined tumors and more favorable baseline characteristics. Partial nephrectomy was performed in 45–52% of young adults in Clemmensen et al [17] and Cakici et al [8], delivering oncologic outcomes comparable to radical nephrectomy while better preserving renal function. Radical nephrectomy was reserved for larger (≥ T2) or centrally located tumors, yet even in these settings oncologic control was excellent when margins were negative. Evidence for systemic therapy is scant: targeted agents and immune checkpoint inhibitors were seldom used in the included cohorts, and observational reports [1, 3] suggest limited activity in non-clear-cell variants.
Limitations of the evidence base
Despite the comprehensive nature of this review, certain limitations must be acknowledged. Variability in study quality resulted in potential selection bias, confounding, and inconsistencies in outcome reporting. The observed heterogeneity in incidence rates, tumor characteristics, and treatment modalities may limit the generalizability of findings. Additionally, small sample sizes in several studies contributed to imprecision, particularly in rare subtypes such as Xp11.2 translocation RCC. Publication bias may also be present, as studies with significant results are more likely to be published. Future research should focus on larger, multicenter studies with standardized methodologies to improve reliability and clinical applicability.
Most included cohorts were retrospective and single-institutional, predisposing to selection bias and unmeasured confounding. Marked between-study heterogeneity was observed for almost every pooled endpoint (I2 > 75%), reflecting regional differences in imaging use, referral thresholds, and surgical practice. Rare subtypes such as Xp11.2 translocation RCC were represented by small case series, widening CIs and limiting precision. Finally, a risk of publication bias exists because smaller studies with null findings are less likely to appear in indexed journals.
Applying the GRADE framework, incidence and histology outcomes were rated low certainty, whereas CSS, recurrence, presentation mode, and stage/grade distributions were downgraded to very low certainty due to serious risk of bias, very serious inconsistency, and imprecision. Consequently, our confidence in the pooled estimates is limited; true effects may differ substantially, and high-quality prospective data are needed to strengthen the evidence base.
Limitations of the review process
Several limitations in the review process should be considered. The search was limited to three databases, PubMed, Scopus, and Web of Science, which may have excluded studies indexed elsewhere. Grey literature, conference abstracts, and unpublished data were not included, which could affect the completeness of the evidence base. Although duplicate removal and screening were performed, automation tools were used during initial screening, which may have excluded relevant studies not captured by algorithmic filters. These factors may influence the comprehensiveness and reproducibility of the review.
Clinical and research implications
The findings of this review have several important clinical and research implications. First, the high rate of incidental tumor detection supports the necessity of increased awareness and the potential benefits of targeted screening strategies in high-risk young adults. Since many renal tumors remain asymptomatic until later stages, improving early detection through routine imaging in high-risk individuals could reduce mortality and enhance treatment efficacy.
Second, given the distinct histological and genetic characteristics of RCC in young adults, treatment strategies should be individualized. Molecular profiling and genetic testing should be incorporated into clinical decision-making, particularly for patients with aggressive or rare tumor subtypes such as Xp11.2 translocation RCC. Identifying genetic markers associated with tumor progression could also aid in the development of targeted therapies.
Future research should prioritize long-term functional outcomes following nephron-sparing surgery, including renal function preservation and quality of life. Additionally, studies should explore the role of emerging adjuvant therapies in improving survival rates for high-risk patients. Multicenter prospective trials are warranted to validate the findings of this review and to bridge existing gaps in knowledge regarding the biological behavior of RCC in young adults.
Conclusion
Renal tumors in adults younger than 45 years remain uncommon (about 0.6 cases per 100,000 person-years), yet their incidence has doubled since 2000. Clear-cell RCC predominates (about 54%), with papillary and chromophobe variants accounting for most of the remainder. In the pooled cohorts, 41% of tumors were detected incidentally and about three-quarters were stage I–II at surgery; these features underpin the excellent 5-year CSS (> 90%) and low recurrence rate (about 4%). Prognosis, however, is heterogeneous: aggressive subtypes such as Xp11.2 translocation RCC or lesions with sarcomatoid differentiation, and any tumor presenting beyond stage II, carry a markedly higher risk of relapse and death.
Management is evolving towards renal preservation. Partial nephrectomy was feasible in roughly half of young adults while maintaining oncologic control. Evidence for systemic therapy in non-clear-cell disease remains sparse, and we need more molecularly guided trials. Given that the current evidence base is of low to very low certainty, future research should prioritize prospective, multicenter cohorts with uniform staging, comprehensive genomic profiling, and extended functional follow-up. Until such data are available, early imaging of high-risk individuals, timely referral to centers experienced in nephron-sparing surgery, and judicious use of emerging targeted or immune-based regimens offer the best opportunity to preserve both survival and quality of life in this distinct patient population.
| Supplementary Material | ▴Top |
Suppl 1. Database Search Strategy and Yield.
Suppl 2. Characteristics of Included Primary Studies.
Suppl 3. Characteristics of Literature Review Studies.
Acknowledgments
The authors thank the National Kidney and Transplant Institute Clinical Trial and Research Unit for administrative support and protocol registry assistance. This study can be found in the National Kidney and Transplant Institute research registry with a protocol number of NKTI-REC-2024-97.
Financial Disclosure
No funding was received for this study.
Conflict of Interest
The authors declare no conflicts of interest.
Informed Consent
Not applicable. This study is a systematic review and meta-analysis of previously published data and did not involve direct interaction with human participants.
Author Contributions
JPR Tadifa: conceptualization, methodology, data curation, writing – original draft. RI De Guzman: data extraction, validation, writing – review and editing. KGM Garcia: supervision, adjudication, final manuscript approval.
Data Availability
The authors declare that data supporting the findings of this study are available within the article and its supplementary materials. Any additional analytic code is available from the corresponding author upon reasonable request.
| References | ▴Top |
- Nayyar R, Khattar M, Dadhwal R, Singh P, Nayak B, Kumar R, Seth A. A large contemporary experience of renal tumors in young: Clinico-pathological profile and long-term survival patterns. Indian J Surg Oncol. 2023;14(1):169-175.
doi pubmed - Daugherty M, Bratslavsky G. Renal cell carcinoma in young patients: a review of recent literature. Curr Urol Rep. 2015;16(2):1.
doi pubmed - Akhavan A, Richards M, Shnorhavorian M, Goldin A, Gow K, Merguerian PA. Renal cell carcinoma in children, adolescents and young adults: a National Cancer Database study. J Urol. 2015;193(4):1336-1341.
doi pubmed - Siemer S, Hack M, Lehmann J, Becker F, Stockle M. Outcome of renal tumors in young adults. J Urol. 2006;175(4):1240-1243; discussion 1243-1244.
doi pubmed - Taccoen X, Valeri A, Descotes JL, Morin V, Stindel E, Doucet L, Joulin V, et al. Renal cell carcinoma in adults 40 years old or less: young age is an independent prognostic factor for cancer-specific survival. Eur Urol. 2007;51(4):980-987.
doi pubmed - Xue Z, Tang S, Ou J, Fang Y, Qiu M, Hong K, Tian X, et al. Clear cell and non-clear cell renal cell carcinoma in young adults: clinicopathological features, survival outcomes and prognostic factors. World J Urol. 2024;42(1):364.
doi pubmed - Abou El Fettouh HI, Cherullo EE, El-Jack M, Al Maslamani Y, Novick AC. Sporadic renal cell carcinoma in young adults: presentation, treatment, and outcome. Urology. 2002;60(5):806-810.
doi pubmed - Cakici MC, Kisa E, Yalcin MY, Efiloglu O, Yucel C, Atis G, Ilbey YO, et al. Influence of border-age on survival of sporadic renal cell carcinoma: young adults versus octogenarians. Int Urol Nephrol. 2020;52(11):2087-2095.
doi pubmed - Aslan R, Taken K, Eryilmaz R. Clinicopathological Features and Survival Data of Localized Renal Masses in Young Adults. Asian Pac J Cancer Prev. 2018;19(11):3233-3236.
doi pubmed - Eggener SE, Rubenstein JN, Smith ND, Nadler RB, Kontak J, Flanigan RC, Waters WB, et al. Renal tumors in young adults. J Urol. 2004;171(1):106-110.
doi pubmed - Mohsin R, Hashmi A, Sultan G, Shehzad A, Mubarak M, Ghazanfar N, Tunio MA, et al. Renal tumors in young adults: a single-center experience from a developing country. Urol J. 2012;9(1):373-380.
pubmed - Lopez JI, Moreno V, Garcia H, Anton I, Robles A, Onate JM, Banos A, et al. Renal cell carcinoma in young adults: a study of 130 cases and a review of previous series. Urol Int. 2010;84(3):292-300.
doi pubmed - Aziz A, May M, Zigeuner R, Pichler M, Chromecki T, Cindolo L, Schips L, et al. Do young patients with renal cell carcinoma feature a distinct outcome after surgery? A comparative analysis of patient age based on the multinational CORONA database. J Urol. 2014;191(2):310-315.
doi pubmed - Xu L, Yang R, Gan W, Chen X, Qiu X, Fu K, Huang J, et al. Xp11.2 translocation renal cell carcinomas in young adults. BMC Urol. 2015;15:57.
doi pubmed - King SC, Pollack LA, Li J, King JB, Master VA. Continued increase in incidence of renal cell carcinoma, especially in young patients and high grade disease: United States 2001 to 2010. J Urol. 2014;191(6):1665-1670.
doi pubmed - Palumbo C, Pecoraro A, Rosiello G, Luzzago S, Deuker M, Stolzenbach F, Tian Z, et al. Renal cell carcinoma incidence rates and trends in young adults aged 20-39 years. Cancer Epidemiol. 2020;67:101762.
doi pubmed - Clemmensen T, Matoso A, Graham T, Lai WS, Rais-Bahrami S, Gordetsky J. Pathologic and clinical characteristics of early onset renal cell carcinoma. Hum Pathol. 2018;74:25-31.
doi pubmed - Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Schunemann HJ. GRADE Handbook for grading quality of evidence and strength of recommendations. Updated 2023. Available from: https://gdt.gradepro.org/app/handbook/handbook.html.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
doi pubmed - Khondker A, Kwong JCC, Chua ME, Kim JK, Chan JYH, Zappitelli M, Brzezinski J, et al. Nephron-sparing surgery for renal cell carcinoma in children and young adults: A systematic review. Urol Oncol. 2023;41(3):137-144.
doi pubmed - Ray S, Jones R, Pritchard-Jones K, Dzhuma K, van den Heuvel-Eibrink M, Tytgat G, van der Beek J, et al. Pediatric and young adult renal cell carcinoma. Pediatr Blood Cancer. 2020;67(11):e28675.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
World Journal of Nephrology and Urology is published by Elmer Press Inc.